Other than the times we notice our battery bars inching towards the red or our screens go silent, we usually don’t think too about how it is our phones actually function. But as a new video from the Reaction YouTube channel shows, there’s a whole host of complex chemical reactions going on both during production and on a daily basis to keep our phones functioning.
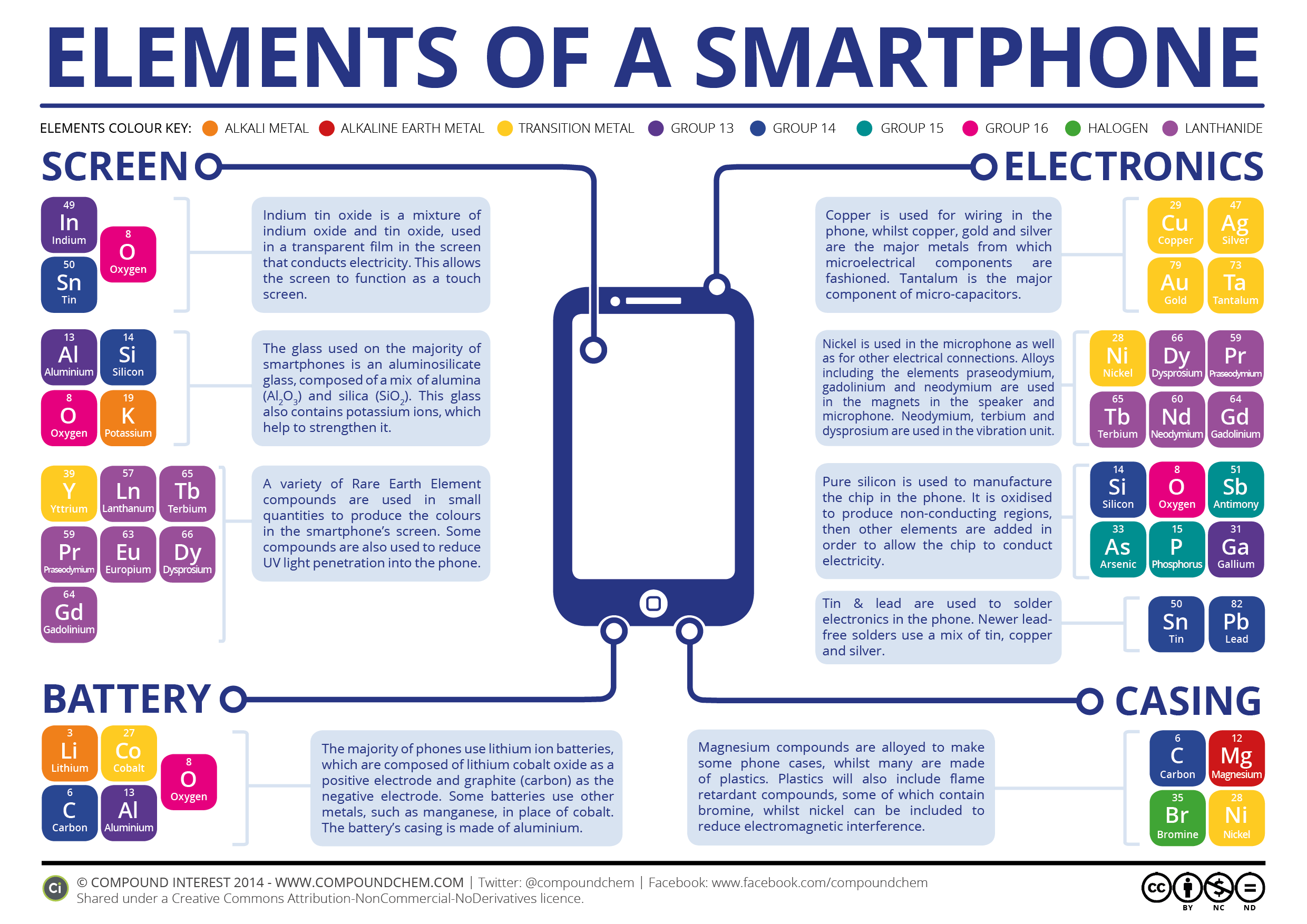
Infographic via Compound Interest.
For instance, you might have heard that the iPhone comes bearing “ion-strengthed glass,” but what does that actually mean. Compound Interest explains:
[Screens] are manufactured mainly from aluminosilicate glass, a mixture of aluminium oxide and silicon dioxide, which is then placed in a hot bath of molten salt. The purpose of this is to allow smaller sodium ions to leave the glass and larger potassium ions to take their place; these take up more room and are pressed together when the glass cools, producing a layer of compressive stress on the glass and increasing its strength and resistance to mechanical damage.
What’s more, in order for your smartphone screen to give off all those pretty colours, it comes loaded with some of the rarest elements on Earth — praseodymium, yttrium, gadolinium, and terbium, to be exact. And injecting the plastic, flame-retardant casing with nickel keeps any electromagnetic interference to a minimum.
It’s a fascinating look at something so many of us take for granted, and you can read more about it over at Compound Interest here. [Reactions]