car-t
-
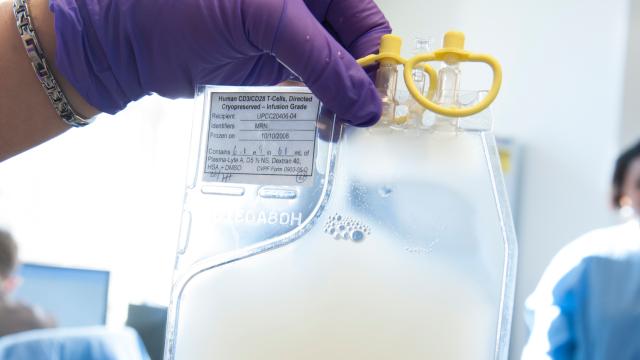
The First US Human CRISPR Trials Could Start Any Day Now
Image: Penn Medicine The first US human trial using CRISPR to treat disease could kick off any day now. The trial, led by the University of Pennsylvania, will use the gene-editing tool to modify immune cells, prompting them to attack three different types of cancer.
-
Did An FDA Panel Really Just Approve The First Gene Therapy Treatment?
This week marked a major milestone in medicine: On Wednesday, a US Food and Drug Administration panel unanimously recommended the approval of the first CAR-T therapy, a pioneering new cancer drug from Novartis that genetically engineers a patient’s own white blood cells to recognise and attack the cancer cells invading their body.