Despite over 200,000 deaths reported worldwide, scientists are only just beginning to understand the impact COVID-19 has on the body. As well as targeting the lungs and the respiratory system, the virus is causing cardiovascular, gastrointestinal and even neurological symptoms.
Early research is also indicating that many of the fatal cases of COVID-19 result from over-activation of a person’s immune system. A better understanding of this phenomenon, often referred to as a “cytokine storm“, could be key in preventing the virus’s deadly effects.
What are cytokines?
The immune system helps to protect us from pathogens, such as bacteria or viruses, when they enter the body.
A host of specialised white blood cells that make up the immune system scour the body’s tissues, seeking to identify pathogens and destroy them. When they identify a pathogen, the immune cells need to trigger inflammation and recruit more immune cells.
The immune response depends on effective cooperation between immune cells. The immune cells communicate via the release of small signalling molecules called cytokines.
Once released, the cytokines trigger localised inflammation. This is a physiological response by the body that aims to destroy the pathogen. Signs of inflammation include redness (increased blood flow), swelling, pain and elevated temperature.
Released cytokines work by binding to receptors found either on nearby cells or even on the same cell that released them. Some cytokines can stimulate further release of cytokines, creating a positive feedback loop and amplifying the inflammation. Often this results in fever, a key hallmark of inflammation.
Importantly, there are many different cytokines. Cytokines are often classified by the type of immune cells that release them, or grouped by their similarity. Each cytokine may trigger a distinct response when binding to a receptor. For example, some increase or decrease inflammation, whereas others could help in recruiting more immune cells or may even damage tissues.
This means that understanding which cytokines are released in response to infection can be as important as knowing the quantities of cytokines released.
Cytokine “storm”
In some cases, the immune system overreacts during an infection, releasing more cytokines than necessary, and progressively recruiting new hordes of activated “angry” white blood cells, which produce even more cytokines. This means a “cytokine storm” is emerging.
Like other signalling molecules (including hormones and neurotransmitters), cytokines can enter the bloodstream. These powerful regulators of inflammation can affect the whole body as seen in sepsis or as a side-effect of some anti-cancer therapies. Large quantities of cytokines can cause widespread (systemic) inflammation that can damage multiple organs.
Some cytokines can also affect the blood vessels, causing a dramatic drop in blood pressure. This limits blood supply and starves the vital tissues and organs ” including the heart, lungs, brain and kidneys ” of oxygen and nutrients.
Cytokines and COVID-19
The exact mechanisms of the “cytokine storm” and their contribution to reported fatalities in COVID-19 are still under investigation.
In many of these patients, the “cytokine storm” caused severe deterioration due to a rapid development of acute respiratory distress syndrome, where the lungs are unable to provide the body’s vital organs with enough oxygen.
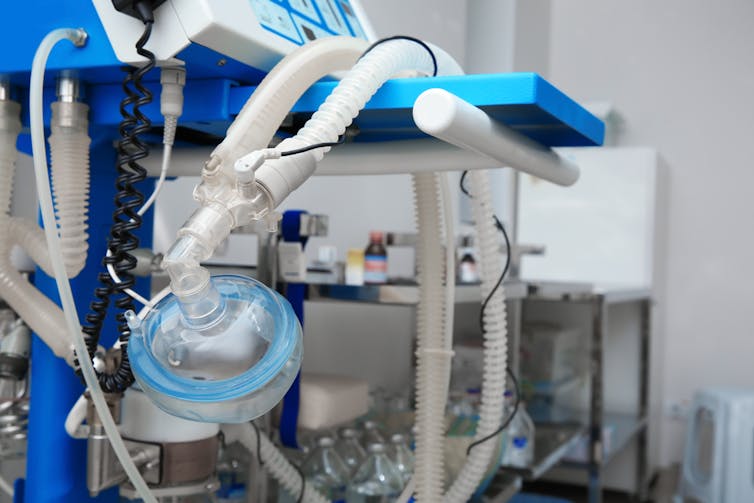
Patients with acute respiratory distress syndrome require ventilators to breathe.
New Africa/ Shutterstock
In such patients, researchers have seen considerable increase of cytokines, particularly those that control migration and activation of macrophages. Macophages are the white blood cells that specialise in removing pathogens and regulating inflammation. If the macrophages migrate to the lungs, as in COVID-19 patients, these cells can cause widespread inflammation resulting in respiratory failure.
Further clinical and immunological analysis of severe COVID-19 cases are necessary for better understanding this infection and identifying treatments.
Current treatments
A number of strategies, both old and new, are already being considered or implemented in an attempt to “calm” the “cytokine storm”.
Treatments look to reduce activation of immune cells by interfering with cytokine production or blocking specific signalling pathways dependent on pro-inflammatory cytokines. The pace of the ongoing pandemic has forced researchers and clinicians to rapidly trial various therapies developed earlier for treatment of other conditions (such as Crohn’s disease and arthritis) caused by an overreactive immune system.
Immunosuppressive interventions, however, could put COVID-19 patients at greater risk as many of them already suffer from a reduction and functional decline in key immune cells. Previous animal studies have also shown that while cytokine neutralisation could prevent development of septic shock, it also weakened their immunity and made them more vulnerable to bacterial infections.
We are witnessing a remarkable acceleration of clinical trials addressing various facets of COVID-19 in the hope of developing more effective treatments. However, efficient targeting of the “cytokine storm” requires further research into the complex physiological and cellular responses.
![]()
Vadim Taraban, Course Leader in Biomedical Sciences, Solent University and Robbie Baldock, Lecturer in Biochemistry, Solent University
This article is republished from The Conversation under a Creative Commons license. Read the original article.