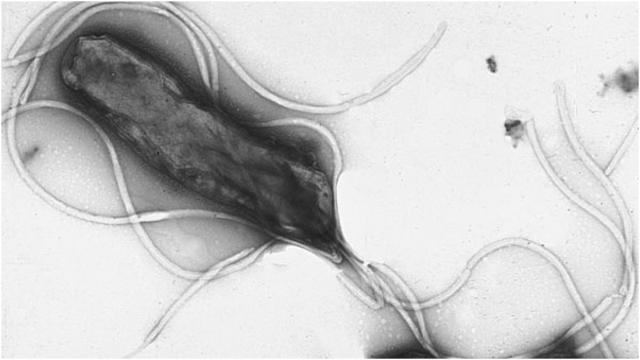
A common bacterial infection that causes stomach ulcers and cancer has gotten increasingly harder to fend off, according to new preliminary research out this week. In Europe, it suggests, strains of the bacteria Helicobacter pylori have become more resistant to the first-line drugs used to kill it over the past 20 years, with resistance rates doubling for at least one antibiotic.
H. pylori loves to live in the human stomach. More than half of the world is colonised with it, though most people don’t fall sick. But occasionally, it can cause acute infections that resemble a typical stomach flu. Over a lifetime, the bacteria can also help cause ulcers that inflame and wear away the lining of the stomach, and this tissue damage increases the risk of stomach cancer. Infamously, scientists assumed for decades that ulcers were solely caused by stress and diet, wrongly believing that microbes could never endure the harsh, acidic environment of our stomachs.
Ulcers caused by H. pylori are routinely treated with antibiotics and drugs that lower stomach acid production. But throughout the years, doctors have noticed that the antibiotics used against it — even when given as a combination therapy — aren’t working as effectively as they once did. Reflecting this concern, the World Health Organisation declared in 2017 that new treatments were urgently needed for strains of H. pylori resistant to the front-line antibiotic clarithromycin.
But according to the authors of this new research, there hasn’t been much work laying out in broad terms just how big of a problem H. pylori resistance has become. So they compared more than 1,200 samples of the bacteria collected from patients by doctors across 18 European countries from 1998 to 2018. Then they tracked how the stomach bug had evolved in its resistance to three commonly employed antibiotics, including clarithromycin.
Overall, they found that only 9.9 per cent of samples in 1998 displayed pre-existing resistance to clarithromycin, a percentage that ballooned to 21.6 per cent by 2018. And in some regions, such as Southern Italy and Croatia, over a third of all H. pylori strains showed clarithromycin resistance. Resistance to the other two drugs, levofloxacin and metronidazole, increased over time as well, with rates of metronidazole resistance being the highest of all three.
The study’s findings are preliminary and were presented this week at the annual conference of the United European Gastroenterology. But they do track with some existing, peer-reviewed research. In 2018, a review similarly found that the average rate of resistance to all three drugs by H. pylori eclipsed 15 per cent in most areas of the world.
Needless to say, it’s not great news.
“With resistance rates to commonly used antibiotics such as clarithromycin increasing at an alarming rate of nearly 1 per cent per year, treatment options for H. pylori will become progressively limited and ineffective if novel treatment strategies remain undeveloped,” lead author Francis Megraud, a professor of bacteriology at the University of Bordeaux in France, said in a release. “The reduced efficacy of current therapies could maintain the high incidence rates of gastric cancer and other conditions such as peptic ulcer disease, if drug resistance continues to increase at this pace.”
There are a few glimmers of hope, at least. The study found that in some countries, such as Denmark and Norway, rates of clarithromycin resistance were still incredibly low, hovering around 5 per cent. These countries have historically been better at keeping antibiotic resistance in check through policies that discourage unnecessary antibiotic use (particularly in livestock). But unless the rest of the world follows suit, and quickly, H. pylori and countless other infections will only become more and more untreatable.