Genetic engineering, researchers hope, will fight disease both by altering the genes of people and by changing the genes of critters known to pass diseases on to us.
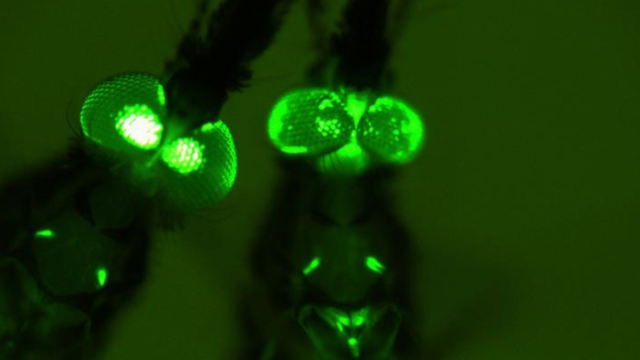
Mosquitoes that have been genetically modified using CRISPR. They glow because of green florescent protein used to track modifications. Photo: Dong Y, et al. (2018)
Take the mosquito, a pest that kills more than half a million people globally each year. It is the primary vector for yellow fever, dengue fever, Zika virus and malaria, among other deadly diseases. Most of those illnesses do not have vaccines or cures. For years, scientists have been tweaking the mosquito’s genome, hoping to interfere with its ability to spread disease to people.
The most high-profile attempts at doing so have involved essentially sterilizing the insects. The British company Oxitec, for example, has been engineering Zika-carrying male Aedes aegypti mosquitoes to carry a lethal gene that kills off any offspring they might have with a wild female, killing off invasive mosquito populations in order to prevent disease.
Now a new paper presents a way to use genetic engineering to tackle perhaps the biggest mosquito-born threat of all: malaria.
Using the buzzy gene-editing technique CRISPR/Cas9, scientists at John Hopkins University knocked out a gene called FREP1 that has been shown to make Anopheles gambiae mosquitoes less susceptible to the parasite that ultimately causes malaria in humans. Several mosquito proteins are involved in the cycle that allows the Plasmodium parasite to make its way to the mosquito’s salivary gland, where it can the spread to bitten humans.
In a study published this week in the journal PLOS Pathogens, researchers showed that using CRISPR to delete the single gene FREP1 successfully interfered with that process.
Malaria is one of the world’s deadliest diseases. Their finding could be a major boon in the fight against a disease that infects hundreds millions of people and kills hundreds of thousands every year.
But first, there are more than a few hurdles. To begin, deactivating the FREP1 in lab-bred mosquitoes also made the mosquitoes less fit, meaning that if those mosquitoes were released into the wild to mate and spread their malaria-resistant DNA, they might not be able to compete for mates against other wild mosquitoes.
Fitness has been a major concern of other projects aimed at altering mosquitoes. Mosquitoes are more fragile than other insects, despite their menacing tendencies. Other insects, for example, are easily blasted with radiation to effectively sterilize them, but when such experiments were done in mosquitoes, they have been too weak to mate — which is a problem, because the idea is to get these mutant mosquitoes to mate in the wild, thus taking mating opportunities away from fitter competitors.
Even if a mosquito appears fine in the lab, there’s no telling how things will play out in the real world. To address this, the study authors are exploring potentially only deactivating that gene in the gut of adult female mosquitoes, hoping the fitness cost will be lower but the resistance still effective.
And while Anopheles gambiae is the primary mosquito vector for malaria in sub-Saharan Africa, there are somewhere around 40 Anopheles subspecies that can transmit malaria to people. There is no telling whether this method will work in other species.
Safety and public opinion will also come into play. It was public opinion, not regulators, that put the kibosh on Oxitec’s trial of its mosquitoes in Key West. The Bill and Melinda Gates Foundation has invested heavily into using a genetic engineering technology called gene drives to tackle malaria, but has described the project as a “long shot,” no doubt due to public objections in addition to the technological hurdles.
Those concerns include unintended consequences that could alter an insect’s behaviour or trigger a troublesome chain reaction in which a whole ecosystem is adversely affected by these altered mosquitos.
Other approaches to fighting malaria have tried genetically modifying bacteria found inside mosquitoes to fight off the parasite, or giving it new genes that help it to not transmit malaria.
But scientists are already preparing for the potential release of modified mosquitoes in malaria-plagued places like Burkina Faso. That day may not exactly be around the corner, but one day genetically altered mosquitoes very well might save thousands of lives.