Carpenter ants of the Brazilian rain forest have it rough. When one of these insects gets infected by a certain fungus, it turns into a so-called “zombie ant” and is no longer in control of its actions. Manipulated by the parasite, an infected ant will leave the cosy confines of its arboreal home and head to the forest floor – an area more suitable for fungal growth. After parking itself on the underside of a leaf, the zombified ant anchors itself into place by chomping down onto the leaf. This marks the ant’s final act. From here, the fungus continues to grow and fester inside the ant, eventually thrusting a stalk through the ant’s head that releases fungal spores. This entire process, from start to finish, can take upwards of ten agonizing days.
We’ve known about zombie ants for quite some time, but scientists have struggled to understand how the parasitic fungus, O. unilateralis (pronounced yu-ni-lat-er-al-iss), performs its puppeteering duties. This fungus is often referred to as a “brain parasite,” but new research published this week in Proceedings of the National Academy of Sciences shows that the brains of these zombie ants are left intact by the parasite, and that O. unilateralis is able to control the actions of its host by infiltrating and surrounding muscle fibres throughout the ant’s body. In effect, it’s converting an infected ant into an externalized version of itself. Zombie ants thus become part insect, part fungus. Awful, right?
To make this discovery, the scientist who first uncovered the zombie ant fungus, David Hughes from Penn State, launched a multidisciplinary effort that involved an international team of entomologists, geneticists, computer scientists, and microbiologists. The point of the study was to look at the cellular interactions between O. unilateralis and the carpenter ant host Camponotus castaneus during a critical stage of the parasite’s life cycle — that phase when the ant anchors itself onto the bottom of leaf with its powerful mandibles.

Ants infected with late stage O. unilateralis infection. (Image: David Hughes/PLOS ONE)
“The fungus is known to secrete tissue-specific metabolites and cause changes in host gene expression as well as atrophy in the mandible muscles of its ant host,” said lead author Maridel Fredericksen, a doctoral candidate at the University of Basel Zoological Institute, Switzerland, in a statement. “The altered host behaviour is an extended phenotype of the microbial parasite’s genes being expressed through the body of its host. But it’s unknown how the fungus coordinates these effects to manipulate the host’s behaviour.”
By referring to the parasite’s “extended phenotype,” Fredericksen is referring to the way that O. unilateralis is able to hijack an external entity, in this case the carpenter ant, and make it a literal extension of its physical self.
For the study, the researchers infected carpenter ants with either O. unilateralis or a less threatening, non-zombifying fungal pathogen known as Beauveria bassiana, which served as the control. By comparing the two different fungi, the researchers were able to discern the specific physiological effects of O. unilateralis on the ants.
Using electron microscopes, the researchers created 3D visualisations to determine location, abundance, and activity of the fungi inside the bodies of the ants. Slices of tissue were taken at a resolution of 50 nanometres, which were captured using a machine that could repeat the slicing and imaging process at a rate of 2,000 times over a 24-hour period. To parse this hideous amount of data, the researchers turned to artificial intelligence, whereby a machine-learning algorithm was taught to differentiate between fungal and ant cells. This allowed the researchers to determine how much of the insect was still ant, and how much of it was converted into the externalized fungus.
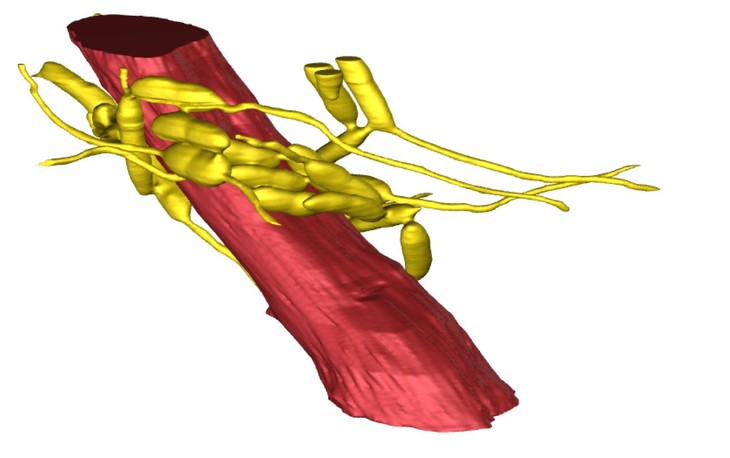
3D reconstruction of an ant mandible adductor muscle (red) surrounded by a network of fungal cells (yellow). (Image: Hughes Laboratory/Penn State)
The results were truly disturbing. Cells of O. unilateralis had proliferated throughout the entire ant’s body, from the head and thorax right down to the abdomen and legs. What’s more, these fungal cells were all interconnected, creating a kind of Borg-like, collective biological network that controlled the ants’ behaviour.
“We found that a high percentage of the cells in a host were fungal cells,” said Hughes in a statement. “In essence, these manipulated animals were a fungus in ants’ clothing.”
But most surprising of all, the fungus hadn’t infiltrated the carpenter ants’ brains.
“Normally in animals, behaviour is controlled by the brain sending signals to the muscles, but our results suggest that the parasite is controlling host behaviour peripherally,” explained Hughes. “Almost like a puppeteer pulls the strings to make a marionette move, the fungus controls the ant’s muscles to manipulate the host’s legs and mandibles.”
As to how the fungus is able to navigate the ant towards the leaf, however, is still largely unknown. And in fact, that the fungus leaves the brain alone may provide a clue. Previous work showed that the fungus may be chemically altering the ants’ brains, leading Hughes’ team to speculate that the fungus needs to the ant to survive long enough to perform its final leaf-biting behaviour. It’s also possible, however, that the fungus needs to leverage some of that existing ant brain power (and attendant sensorial capabilities) to “steer” the ant around the forest floor. Future research will be required to turn these theories into something more substantial.
“This is an excellent example of how interdisciplinary research can drive our knowledge forward,” Charissa de Bekker, an entomologist at the University of Central Florida not affiliated with the new study, told Gizmodo. “The researchers used cutting-edge techniques to finally confirm something that we thought to be true but weren’t sure about: that the fungus O. unilateralis does not invade or damage the brain.”
de Bekker says this work confirms that something much more intricate is going on, and that the fungus might be controlling the ant by secreting compounds that can work as neuromodulators. And importantly, data gleaned from the fungal genome points to this conclusion as well.
“This means the fungus might produce a wealth of bioactive compounds that could be of interest in terms of novel drug discovery,” said de Bekker. “I am, thus, very excited about this work!”
An authority on the zombie ant fungus herself, de Bekker also released new research this week. Her new study, published in PLOS One and co-authored with David Hughes and others, looked into the molecular clock of the Ophiocordyceps kimflemingiae fungus (a recently named species of the O. unilateralis complex) to see if the daily rhythms, and thus biological clocks, are an important aspect of the parasite-host interactions studied by biologists.
“In addition to confirming that the fungus indeed has a molecular clock, we found that this results in the daily oscillation of certain genes,” de Bekker told Gizmodo. “While some of them are active during the day-time, others are active during the night-time. Interestingly, we found that the fungus especially activates genes encoding for secreted proteins during the night-time. These are the compounds that possibly interact with the host’s brain! The fungus, therefore, does not just release bioactive compounds to manipulate behaviour, but there seems to be a precise timing to it as well.”
There’s clearly still lots to learn about this insidious parasite and how it hijacks its insectoid hosts, but as these recent studies attest, we’re getting steadier closer to the answer — one that’s clearly disturbing in nature.
