Like humans, gorillas and chimpanzees can get infected by Ebola. To protect our closest relatives from this dreaded disease, and to prevent the virus from spilling over into human populations, scientists have now developed an oral vaccine to combat Ebola in the wild. It sounds very promising, but the researchers have run into legal, political and ethical obstacles that could make it difficult to push the research further.
Image: USAID Africa Bureau.
Ingestible vaccines are nothing new, but this marks the first time that scientists have developed one specifically for the purpose of protecting and preserving wild apes. This new vaccine, should it ever be put to use, would indirectly help humans by reducing Ebola’s opportunity to take root in wild animal populations, from where it could spread to humans. But in order for this to happen, and to allow scientists the opportunity to push this work further, we may have to rethink animal experimentation — particularly when the animals themselves stand to benefit.

In addition to having a shared ancestry, humans and apes both have to contend with the Ebola virus. Over the past three decades, the disease has killed about a third of the world’s gorillas and countless chimpanzees. Ebola threatens wild primates that are already at risk, be it from habitat loss or rampant poaching.
The virus, like so many other diseases, can jump from species to species — in this case, fruit bats (perhaps the most prolific spreader of the disease), monkeys, forest antelope, porcupines and various apes. Suffice to say, the fewer wild animals infected by the virus, the better.
Thankfully, Ebola can be curbed with vaccines, and there have been several recent developments on that front. The challenge, however, is delivering the vaccine to vulnerable populations in the wild. Conservationists have experimented with hypodermic darts, but it has proven difficult to target animals who live in thick tropical forests and fear humans, like so many primates do.
A potential solution to this problem comes in the form of ingestible vaccines. And in fact, conservationists have had great success with oral vaccines in the fight against rabies. Since 1983, more than 250 million baits containing a rabies vaccine have been distributed in Europe, resulting a 75 per cent decrease of the disease in wild animal populations. Similar effects have been seen in North America.
With this idea in mind, researchers from the University of Cambridge and Thomas Jefferson University embarked on a project to see if a similar oral vaccine could be developed to protect wild apes from Ebola. Using a promising Ebola vaccine called filorab1, and combining it with an inactive version of the rabies virus, the researchers were able to create an oral version of the vaccine. The disabled rabies virus allows the Ebola vaccine to pass more easily into the bloodstream when ingested, triggering a robust immune response.
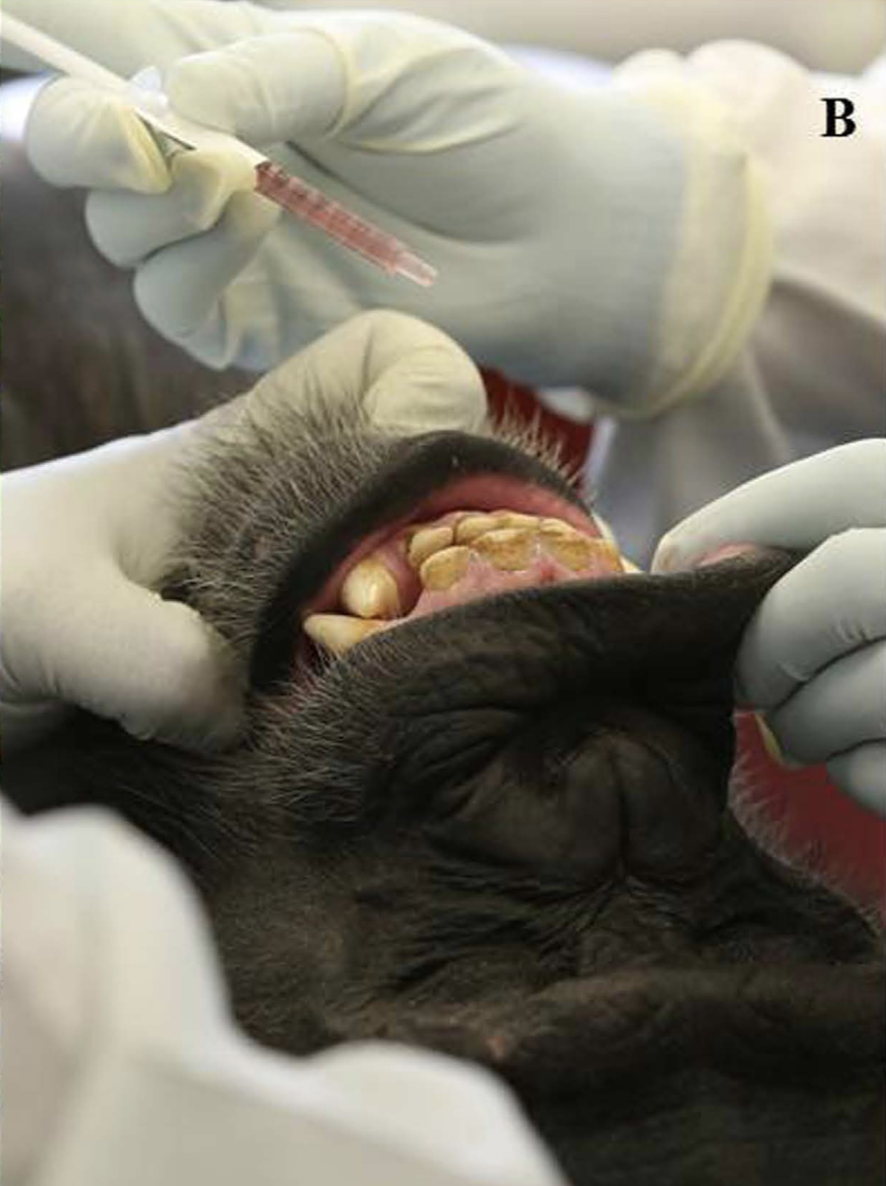
This oral vaccine was tested on 10 captive chimps who were being studied at the New Iberia Research Center in Louisiana. The researchers monitored the chimps for nearly a month, assessing the animals for any side effects and measuring the amounts of anti-Ebola antibodies in their blood. During the 28-day trial, the researchers watched as the antibodies increased in volume, indicating that the primates had developed an immunity to the virus. No serious side effects were recorded.
The scientists say this represents a new potential strategy for combating Ebola, one that could protect both apes and ourselves from the disease. The oral vaccine could eventually be injected into tasty baits that would be scattered throughout ape habitats, inoculating the population over time. The researchers say a similar oral vaccine could be developed for other diseases that plague wild apes, such as anthrax. This research now appears in Scientific Reports.
It’s important to note, however, that the chimps were never exposed to the Ebola virus after being immunised, so the researchers aren’t actually certain that it works.
The study was not permitted to take that next step — exposing the chimps to the virus — on account of a modification made to the Endangered Species Act in 2015, which made it illegal to perform biomedical research on chimpanzees in the United States. That said, trials on captive chimps are still technically legal in the US if the experiments help the species as a whole. According to the 2015 provisions:
Permits will be issued for these activities only for scientific purposes that benefit the species in the wild, or to enhance the propagation or survival of chimpanzees, including habitat restoration and research on chimpanzees in the wild that contributes to improved management and recovery.
The study’s lead researcher, Peter Walsh, believes his study applies, and that his research should be allowed to continue to the next stage where chimps actually get exposed to the virus.
“I believe we have an extremely strong case,” he told Gizmodo. “However, all of the biomedical facilities that hold chimps are ‘retiring’ their chimps to sanctuaries and don’t want to apply for a permit.”
He says sanctuaries are philosophically opposed to vaccination, and most don’t have the personnel or facilities to conduct trials safely and rigorously. Some zoos have the facilities and personnel, but Walsh says they don’t want to touch this sort of research for fear of being attacked by animal welfare advocates.
“The conditions chimps experience during trials are clearly not what you want over the long term but they are very far from torture,” he says. “A minority of chimps behave like young children when they get vaccinated, screaming and carrying on. This causes a stress level comparable to college students studying for exams. Otherwise, the major downside is boredom. But this is only for a month or two and the vast majority of their time is spent in social groups with outdoor areas.” Obviously, actually exposing chimps to Ebola is a much more ethically fraught thing to do.

Image: Jose McKenna/Flickr
But until more tests can be done, Walsh’s new oral vaccine isn’t going anywhere. Park managers in Africa won’t let the researchers use a vaccine on endangered apes in the wild until it has been tested on captive apes that have been directly exposed to Ebola, or humans for that matter. Park managers are worried that the infection caused by the vaccine would escape immune control and sicken or kill gorillas or other animals who have eaten the baits. “However, we chose this Ebola vaccine precisely because the rabies vaccine on which it is based has an astoundingly good safety record in use to virtually eradicate fox rabies from Western Europe,” said Walsh.
But Wash says there are other challenges as well.
“Staff from… the Wildlife Conservation Society have been trying to block me from [deploying our oral vaccine in the wild],” he told Gizmodo. “They say it is because oral vaccination is risky. Based on the… facts, I say it is not and feel that they are attempting to block me because of turf politics rather than scientific evidence.”
Looking ahead, the researchers would like to find an oral bait that gorillas will readily eat. Failing that, and without the ability to conduct further tests on the chimps, he says conservationists may have to rely on vaccination darts or some kind of spray. Ultimately, however, Walsh says it’s important to understand the overriding goal of this research.
“This is not just about Ebola, he says. “Infectious disease is one of the big three factors driving our closest relatives rapidly towards extinction — the other two being commercial bushmeat hunting and habitat loss. This oral vaccine trial is an important step in the process of developing and oral delivery system that will allow us to vaccinate and treat wild apes against the many other disease threats.”
