Researchers from South Korea have engineered a strain of bacteria that infiltrates tumours and fools the body’s immune system into attacking cancer cells. In experiments, the modified bacteria worked to reduce cancer in mice, raising hope for human trials.
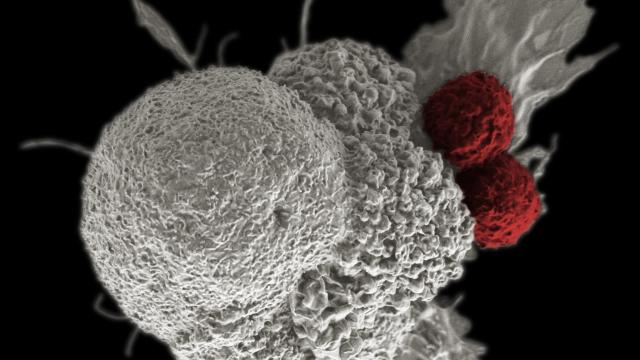
A cancer cell (white) being attacked by immune cells (in red). A new bacterial cancer therapy gives a boost to this natural anti-cancer attack. (Image: NIH)
In a study published today in Science Translational Medicine, a research team led by biologists Joon Haeng Rhee and Jung-Joon Min from Chonnam National University in South Korea describe a new immunotherapy in which a bioengineered strain of Salmonella is converted into a biological version of the fabled Trojan Horse. Once inside an unsuspecting tumour, the modified bacteria transmits a signal that triggers nearby immune cells into launching an attack on the malignant cells.
In preliminary tests, the technique shrunk tumours in more than half of the mice who received injections of the commandeered bacteria. It’s preliminary, but the researchers are hopeful that this form of immunotherapy will be both safe and effective in humans.
Bacterial cancer therapy dates back to 1893 when surgeon William B. Coley noticed that recurrent tumours of connective tissue, called sarcoma tumours, disappeared after patients became infected with Erysipelas bacterium. This led him to develop a therapy, now called “Coley’s toxins”, that utilises various bacterial strains to fight cancer. This line of cancer research went into hibernation, however, once surgical and chemical therapies emerged.
But since 2008, bacterial cancer therapy has been going through a bit of a renaissance. Some success in this area has been reported, particularly when scientists use modified Salmonella to deliver various therapeutic agents to the body, such as genes and anti-cancer medicines. Bacteria have a natural ability to home in on tumours, making them excellent cargo ships. Unfortunately, all pre-existing bacterial cancer therapies require multiple injections of the microbes, and relapses are common.
In an effort to develop a better method, Rhee and Min took a strain of Salmonella typhimurium and made it 10,000 times less toxic than normal, while retaining the bacterium’s ability to stay alive and carry therapeutic cargo. And unlike previous efforts, the modified bacteria weren’t designed to deliver medicines per se; instead, they were engineered to deliver an important message to nearby immune cells — a message that says, “Attack this tumour!”

Credit: Chonnam National University
Specifically, the Salmonella bacteria is genetically modified to secrete a foreign protein known as flagellin (FlaB). This protein, found in an aquatic microbe called Vibrio vulnificus, is the building block of flagellum — the lash-like appendage that allows microorganisms to swim around. Since vertebrate animals, including humans, don’t have a flagellum, this protein is foreign to our cells. When voracious white blood cells known as macrophages detect the presence of these foreign proteins, they immediately sense danger and spring into action.
Macrophages are like microscopic Roombas, vacuuming anything that doesn’t look like it’s supposed to be there, including bits of cellular debris, unfamiliar substances, viruses, unwanted bacteria and, importantly, cancer cells. But macrophages don’t always see tumours as a threat, owing to the presence of identifiable markers, such familiar proteins associated with healthy cells. The modified bacteria, parked inside a tumour with its alien cargo, basically tricks the immune cells into launching an attack (that is, triggering an immune response). The macrophages then happily go about devouring the malignant cells.
The researchers tested their Trojan Horse bacteria in mice with colon cancer. Three days after the injections, the bacteria inside the tumours were 10,000 times more abundant than those found in the mice’s vital organs. The macrophages then quickly went to work, causing the tumours to shrink below detectable limits in more than half of the mice.
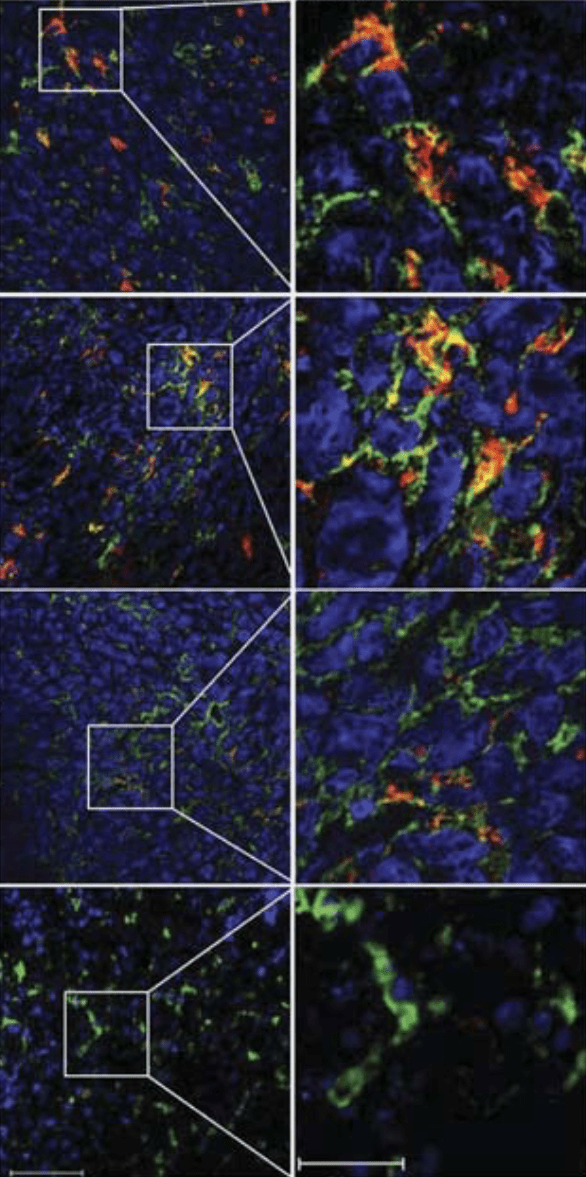
Microscopic images show the macrophage immune cells (the colourful splotches) proliferating and working in cancerous cells. (Image: Jin Hai Zheng et al., 2017)
“We [documented the] total eradication of tumours in approximately 60 per cent of treated subjects,” explained Rhee and Min in an interview with Gizmodo. “The remaining 20 per cent of the animals remained stable,” meaning they didn’t die, “though tumours were reduced in size.”
Importantly, the modified FlaB-expressing bacteria was shown to be non-toxic, and it didn’t invade non-cancerous tissues in the rodents. Instead, Salmonella, armed with FlaB, shrank the tumours, prolonged the survival of the mice, and prevented new growths from re-appearing in mouse models of human colon cancer.
“We did not use any extra medication or chemotherapy,” said Rhee and Min, adding that their bacterial therapy could be combined with other anti-cancer techniques, such as radiation or chemotherapy.
Should this form of immunotherapy reach the clinical stage (and that’s still a big if — mice studies can be notoriously unreliable), the researchers don’t see regular injections as being appropriate.
“The bacteria could be injected repeatedly if required,” noted the researchers. “But we do not encourage multiple repeated therapies since the human immune system will build up an antibacterial immune response after repeated administration of the same bacteria.”An antibacterial response would prevent Salmonella from proliferating at the tumour site, meaning the tumour would no longer be marked for destruction.
To cope with this problem, the researchers are planning to use different strains with distinct biological markers should repeated injections be required in some instances.
It’s too early to tell if this treatment will be effective and safe in humans, but these early results are encouraging. Rhee and Min say their technique should work for other cancers, including breast cancer, glioma, melanoma and lung cancer. “This is more like proof of concept study,” said the researchers.
“We are planning comprehensive preclinical tests in nearest future. If we find right partners that grant reasonable funding, clinical trials could hopefully be started very soon.”