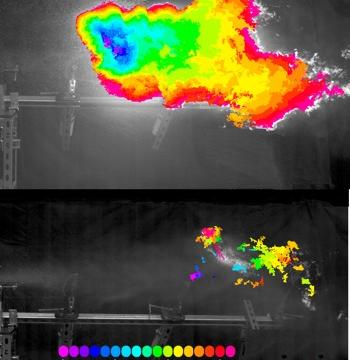
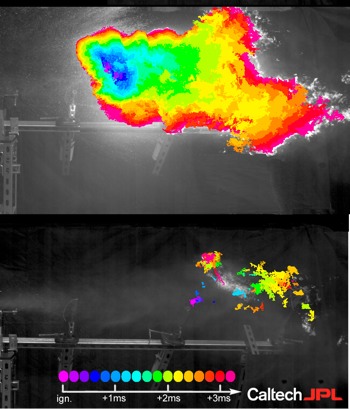
Jet aeroplanes load up with tens of thousands of gallons of fuel for transcontinental flights, which can lead to massive explosions in a crash or terrorist attack. But adding “molecular velcro” to fuel can dramatically reduce its volatility, or explosiveness.
You can see the difference between the two in the above high-speed video. The untreated fuel ignites and generates a massive fireball; the treated fuel does not.
The key is a new kind of ultra-long polymer molecule that Caltech chemical engineer Julia Kornfeld calls “megasupramolecules.” Polymers are just very long chains of repeating blocks of smaller molecules, and the “megasupra” version has the added feature of being capped on either end by blocks that act a lot like velcro. This makes them ideal for controlling the “misting” of jet fuel.
Misting is a good thing when it comes to evenly distributing sprays of fuel for easy ignition in an jet engine. But it becomes a major liability in a crash.
That’s where megasupramolecules can help, by adding them to fuel, like diesel. Most of the time, these supramolecules hang about in a tightly coiled configuration, but the force of an impact makes them stretch out to prevent the breakup of fuel droplets and reduce misting, thereby controlling the size of an explosion and/or slowing the speed at which it ignites.
The idea for these megasupramolecules dates back to the late 1970s, in the wake of one of the deadliest aeroplane crashes in history. In 1977, two Boeing 747s collided on the runway at Tenerife and exploded, killing 583 of the 644 passengers on board. So scientists came up with the idea of adding ultra-long polymers to fuel to decrease the intensity of post-crash explosions.
It seemed like a promising avenue of research, until a disastrous crash test in 1984. Instead of reducing the intensity of the explosion, the test aircraft was consumed by a massive fireball. That failure — not to mention the critical media coverage — effectively squelched interest in developing this application for ultra-long polymers further.
But then 9/11 happened, and JPL researcher Virendra Sarohia approached Kornfeld about reviving the idea of designing a special polymer to help with mist control of jet fuel.
In 2006, one of her grad students, Ameri David, succeeded in devising a computer model for a megasupramolecule that seemed to hit the sweet spot: providing all the benefits of ultra-long polymers as a fuel additive, while addressing some of the shortcomings — namely, a tendency to break apart when flowing through pumps, pipes, and filters. The megasupramolecules, on contrast, can reconnect if they break apart, thanks to that molecular velcro at each end.
It fell to Ming-Hsin “Jeremy” Wei to actually build the structures. The tricky part was fashioning the molecular velcro and placing in the just the right spot on the long polymer chains. JPL chemistry Simon Jones helped him devise better methods for producing longer chains with this structure, although none will be sufficient to scale up production to mass commercial levels if it’s approved s a fuel additive. “My goal is to develop a reactor that will continuously produce the polymer,” Wei said in a press release, adding that he thinks he can do this within the year.
Here’s Kornfeld talking about some of the more promising applications for this new fuel additive. You can see more videos here.
Videos and image: Caltech/JPL
Reference:
Wei, Ming-Hsin et al. (2015) “Megasupramolecules for safer, cleaner fuel by end association of long telechelic polymers,” Science, 350 (6256): 72-75.