CRISPR, a new genome editing tool, could transform the field of biology — and a recent study on genetically-engineered human embryos has converted this promise into media hype. But scientists have been tinkering with genomes for decades. Why is CRISPR suddenly such a big deal?
The short answer is that CRISPR allows scientists to edit genomes with unprecedented precision, efficiency, and flexibility. The past few years have seen a flurry of “firsts” with CRISPR, from creating monkeys with targeted mutations to preventing HIV infection in human cells. Earlier this month, Chinese scientists announced they applied the technique to nonviable human embryos, hinting at CRISPR’s potential to cure any genetic disease. And yes, it might even lead to designer babies. (Though, as the results of that study show, it’s still far from ready for the doctor’s office.)
In short, CRISPR is far better than older techniques for gene splicing and editing. And you know what? Scientists didn’t invent it.
CRISPR/Cas9 comes from strep bacteria…
CRISPR is actually a naturally-occurring, ancient defence mechanism found in a wide range of bacteria. As far as back the 1980s, scientists observed a strange pattern in some bacterial genomes. One DNA sequence would be repeated over and over again, with unique sequences in between the repeats. They called this odd configuration “clustered regularly interspaced short palindromic repeats,” or CRISPR.
This was all puzzling until scientists realised the unique sequences in between the repeats matched the DNA of viruses — specifically viruses that prey on bacteria. It turns out CRISPR is one part of the bacteria’s immune system, which keeps bits of dangerous viruses around so it can recognise and defend against those viruses next time they attack. The second part of the defence mechanism is a set of enzymes called Cas (CRISPR-associated proteins), which can precisely snip DNA and slice the hell out of invading viruses. Conveniently, the genes that encode for Cas are always sitting somewhere near the CRISPR sequences.
Here is how they work together to disable viruses, as Carl Zimmer elegantly explains in Quanta:
As the CRISPR region fills with virus DNA, it becomes a molecular most-wanted gallery, representing the enemies the microbe has encountered. The microbe can then use this viral DNA to turn Cas enzymes into precision-guided weapons. The microbe copies the genetic material in each spacer into an RNA molecule. Cas enzymes then take up one of the RNA molecules and cradle it. Together, the viral RNA and the Casenzymes drift through the cell. If they encounter genetic material from a virus that matches the CRISPR RNA, the RNA latches on tightly. The Cas enzymes then chop the DNA in two, preventing the virus from replicating.
There are a number Cas enzymes, but the best known is called Cas9. It comes from Streptococcus pyogenes, better known as the bacteria that causes strep throat. Together, they form the CRISPR/Cas9 system, though it’s often shortened to just CRISPR.
Top image: Screenshot from this MIT video explaining CRISPR
It is a more precise way of editing the genome…
As this point, you can start connecting the dots: Cas9 is an enzyme that snips DNA, and CRISPR is a collection of DNA sequences that tells Cas9 exactly where to snip. All biologists have to do is feed Cas9 the right sequence, called a guide RNA, and boom, you can cut and paste bits of DNA sequence into the genome wherever you want.
DNA is a very long string of four different bases: A, T, C, and G. Other enzymes used in molecular biology might make a cut every time they see, say, a TCGA sequence, going wild and dicing up the entire genome. The CRISPR/Cas9 system doesn’t do that.
Cas9 takes guide sequences that are about 80 bases long, so they can be highly tailored to a specific gene. All you have to do is design a target sequence using an online tool and order the guide RNA to match. It takes no longer than few days for the guide sequence to arrive by mail. You can even repair a faulty gene by cutting out it with CRISPR/Cas9 and injecting a normal copy of it into a cell. Occasionally, though, the enzyme still cuts in the wrong place, which is one of the stumbling blocks for wider clinical use.
…and way more efficient…
Mice whose genes have been altered or “knocked out” (disabled) are the workhorses for biomedical research. It can take over a year to establish new lines of genetically-altered mice with traditional techniques. But it takes just few months with CRISPR/Cas9, sparing the lives of many mice and saving time.
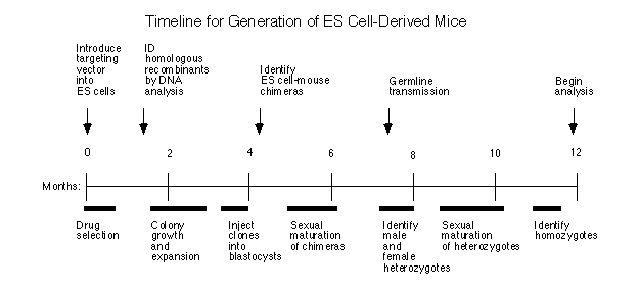
Traditionally, a knockout mouse is made using embryonic stem (ES) cells. Researchers inject the altered DNA sequence into mouse embryos, and hope they are incorporated through a rare process called homologous recombination. Some of first generation mice will be chimeras, their bodies a mixture of cells with and without the mutated sequence. Only some of the chimeras will have reproductive organs that make sperm with mutated sequence. Researchers breed those chimeras with normal mice to get a second generation, and hope that some of them are heterozygous, aka carrying one normal copy of the gene and one mutated copy of the gene in every cell. If you breed two of those heterozygous mice together, you’ll be lucky to get a third generation mouse with two copies of the mutant gene. So it takes at least three generations of mice to get your experimental mutant for research. Here it is summarised in a timeline:
But here’s how a knockout mouse is made with CRISPR. Researchers inject the CRISPR/Cas9 sequences into mouse embryos. The system edits both copies of a gene at the same time, and you get the mouse in one generation. With CRISPR/Cas9, you can also alter, say, fives genes at once, whereas you would have to had to go that same laborious, multi-generational process five times before.
CRISPR is also more efficient than two other genome engineering techniques called zinc finger nuclease (ZFN) and transcription activator-like effector nucleases (TALENs). ZFN and TALENs can recognise longer DNA sequences and they theoretically have better specificity than CRISPR/Cas9, but they also have a major downside. Scientists have to create a custom-designed ZFN or TALEN protein each time, and they often have to create several variations before finding one that works. It’s far easier to create a RNA guide sequence for CRISPR/Cas9, and it’s far more likely to work.
…and can be used in any organism
Most science experiments are done on a limited set of model organisms: mice, rats, zebrafish, fruit flies, and a nematode called C. elegans. That’s mostly because these are the organisms scientists have studied most closely and know how to manipulate genetically.
But with CRISPR/Cas9, it’s theoretically possible to modify the genomes of any animal under the sun. That includes humans. CRISPR could one day hold the cure to any number of genetic diseases, but of course human genetic manipulation is ethically fraught and still far from becoming routine.
Closer to reality are other genetically modified creatures — and not just the ones in labs. CRISPR could become a major force in ecology and conservation, especially when paired with other molecular biology tools. It could, for example, be used to introduce genes that slowly kill off the mosquitos spreading malaria. Or genes that put the brakes on invasive species like weeds. It could be the next great leap in conserving or enhancing our environment — opening up a whole new box of risks and rewards.
With the recent human embryo editing news, CRISPR has been getting a lot of coverage as a future medical treatment. But focusing on medicine alone is narrow-minded. Precise genome engineering has the potential to alter not just us, but the entire world and all its ecosystems.