Ever found yourself eyeing stock lab alcohols and wondering if any of them would be good for a quick buzz without, you know, potentially killing you? Josh Velson, a chemical engineering consultant who works with bio and petrochemicals, has your answer.
The short answer is many — if blindness is the only thing you have to worry about. But let’s break it down.
Methanol
Ah yes, the classic eye-killer. In the body, it is metabolised first to formaldehyde and then to formic acid, which attacks the optic nerve. The best “cure” for accidental (or for that matter, intentional) methanol poisoning is competitive inhibition with ethanol, also known as getting drunk the regular way. Let’s skip over this one, because the others are much more interesting.
n-Propanol
Propanol is a CNS depressant on the order of 4 times more powerful than ethanol. The toxic dose is actually also about 4 times less than ethanol. In the body, it is metabolized to propionaldehdye and thence to propionic acid, which can then be safely disposed of – though it might cause some gastrointestinal distress. So far, so good, right? No dead optic nerves, no ridiculously high oral toxicity… well, don’t get your party hat out yet.
Laboratory grade n-propanol (and there is basically no other significant grade, although you can get natural n-propanol from distilling biological fusel oils) is going to be produced from the OXO process by the hydroformylation of ethylene. There are significant contaminants from that process. I’ve highlighted a choice selection below:
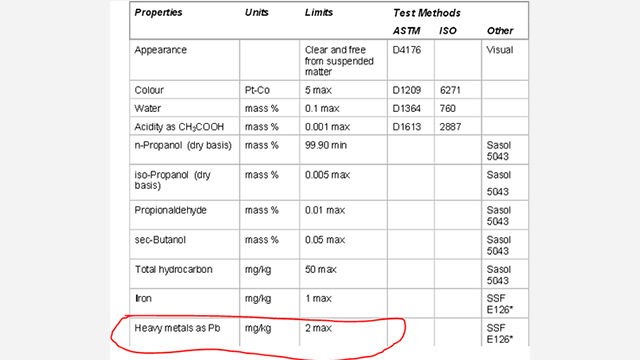
Oh boy. You can figure out what you want about the other alcohols below, but note the “heavy metals as lead” item. Yep, you will have some heavy metals, (assumed to be lead), left over from the OXO process catalysts. The maximum allowable level is 2 mg/kg, also known as 2 parts per million. You know what the EPA says the safe concentration of lead is? Zero. There is no safe concentration. In fact, if some tests consistently show that any drinking water supply has above 15 parts per billion, then water utilities have to start replacing pipes. This is two orders of magnitude above that.
Based on the catalyst type, I would actually suspect that cobalt is the main risk. Long-term cobalt exposure at low levels historically led to a peculiar form of heart problem associated with drinking beer that had been used with cobalt-containing antifoam compounds.
So uh, yeah, it’ll get you really drunk really fast, but you get a risk of lead or other heavy metal poisoning. But you’re hardcore*, right? You could use it to spike some punch the next time you feel like getting your husband gored by a wild boar, or you could just drink it anyway, because hey, YOLO, right?
* Hardcore: “It was really stupid, but I survived.”
Isopropanol
Similar to n-propanol, it’s also a CNS depressant, and has a similar level of oral toxicity – about twice that of ethanol. No word on how much it takes to get you smashed, unfortunately, but what I do know is that in the body it metabolizes to acetone. So, uh, yeah, you might as well be drinking nail polish remover. Within 15 minutes, your breath will smell as if you have anyway! Lethal doses are 100 to 250 mL.
Depending on the source, you may be exposed to a wide variety of contaminants. There are no specs for specific contaminants in 99% grade, but azeotropic mixtures are likely to have sulfur and iron containing compounds. Yummy.
It also has serious reproductive toxicity effects, and, true to any alcohol, has some serious detrimental effects on your liver and kidneys.
Needless to say, this entire answer has been tongue-in-cheek. But it wouldn’t be the first time people tried to get drunk by drinking solvents. Records have indicated that people regularly tried drinking kerosene… and Russian Bears have apparently learned to sniff jet fuel until they pass out.
[National Library of Medicine HSDB Database, Isopropyl Alcohol Poisoning, Page on Colonialchemicals, Basic Information About Lead Regulations (US EPA), Cobalt (Clinical Toxicology), Isopropyl Alcohol – Kirk-Othmer Encyclopedia of Chemical Technology – Logsdon – Wiley Online Library, Page on Dow, Russian bears who are so addicted to aviation fuel they sniff it until they pass out]
Picture: Shutterstock/Luis Louro
This post was republished with permission from Josh Velson, a chemical engineering consultant for bio and petrochemicals. It originally appeared on Quora.
