Shortly after the covid-19 pandemic shut the world down last year, many wearables makers and researchers announced studies to see if their technology could potentially help detect the virus before it spread. Ava, a fertility-tracking wearables company, launched a particularly ambitious study in which 2,000 Liechtenstein citizens wore the company’s trackers to see if the device could pick up covid symptoms before they appeared. Now Ava has announced that the company is launching another study to see if its tracker can detect pre-symptomatic covid-19 in real-time. But with the vaccine now more widely available and an end to lockdowns in sight, what does continuing this kind of research really mean?
One of the biggest issues with covid-19 detection is the fact that a person can spread the virus before they even realise they’re sick. Wearable devices, which are capable of collecting data 24/7 over an extended period of time, seemed like a natural platform for researchers looking to learn more about covid-19 and stem its spread. Ava’s first collaboration with Liechtenstein researchers found that an algorithm could accurately detect 70% of covid-19 positive infections up to seven days before symptoms appeared.
That’s cool, but this capability isn’t unique to Ava. The West Virginia University Rockefeller Neuroscience Institute (RNI) released preliminary research several months ago that it could detect covid-19 symptoms up to three days before they appeared with a 90% accuracy rate. (The sample size was very small.) Whoop also published a peer-reviewed paper saying its recovery tracker could be used to detect covid-19 infections based on dips in users’ respiration rate during sleep. Hell, even Fitbit is doing its own study. The thing is, most of these studies were trying to see if early detection was feasible. There’s a gap between “this thing could work” to “this is how it will be used in real life.”
[referenced id=”1671550″ url=”https://gizmodo.com.au/2021/02/apple-watch-can-detect-covid-19-a-week-early-study-finds/” thumb=”https://gizmodo.com.au/wp-content/uploads/2021/02/12/lnoim278zel9pwyjhuwt-300×169.png” title=”Apple Watch Can Detect Covid-19 a Week Early, Study Finds” excerpt=”In the early days of the pandemic, dozens of researchers launched studies trying to see whether wearables of all sorts could potentially identify covid-19 before symptoms began. In one such study, researchers from Mount Sinai discovered that the Apple Watch was capable of detecting the onset of covid-19 up to…”]
What makes Ava’s new study notable is the fact that it’s taking the algorithm it built and testing how effective it is in real-time and in the real world. Basically, this is the next step to getting this sort of predictive tech out from the lab and onto consumers’ wrists.
For the new study, Ava plans on providing more than 20,000 participants of all genders in both general and high-risk populations with a tracker. They’ll then wear the Ava tracker for up to nine months at night while sleeping, and in the morning can see an AI-generated health indicator: “no change,” “minor changes so stay home and self-isolate,” and “changes that might signal covid-19.” Asymptomatic participants will also be given a diagnostic test, should the app identify them as potentially ill.
Another interesting tidbit is that because Ava is a fertility tracker, the company trained the algorithm on data from nearly 500 women who had reported infection in the app. That data was then extrapolated to non-female subjects. This is significant, because for the overwhelming majority of medical history, it’s been the other way around.
“While historically medical research has relied on data and research using male participants then generalized to women, we are flipping the script at Ava — understanding women’s physiology first, then adapting algorithms to generalise to the whole population,” Ava cofounder and CEO Lea von Bidder said in a statement.
Still, even if this ends up being an effective trial, the study won’t end until November. By that point, most people should have received a covid-19 vaccine. Unfortunately, this is one instance where necessary, rigorous clinical testing sometimes isn’t fast enough to deal with current events as they’re happening — even if there’s regulatory help expediting the process.
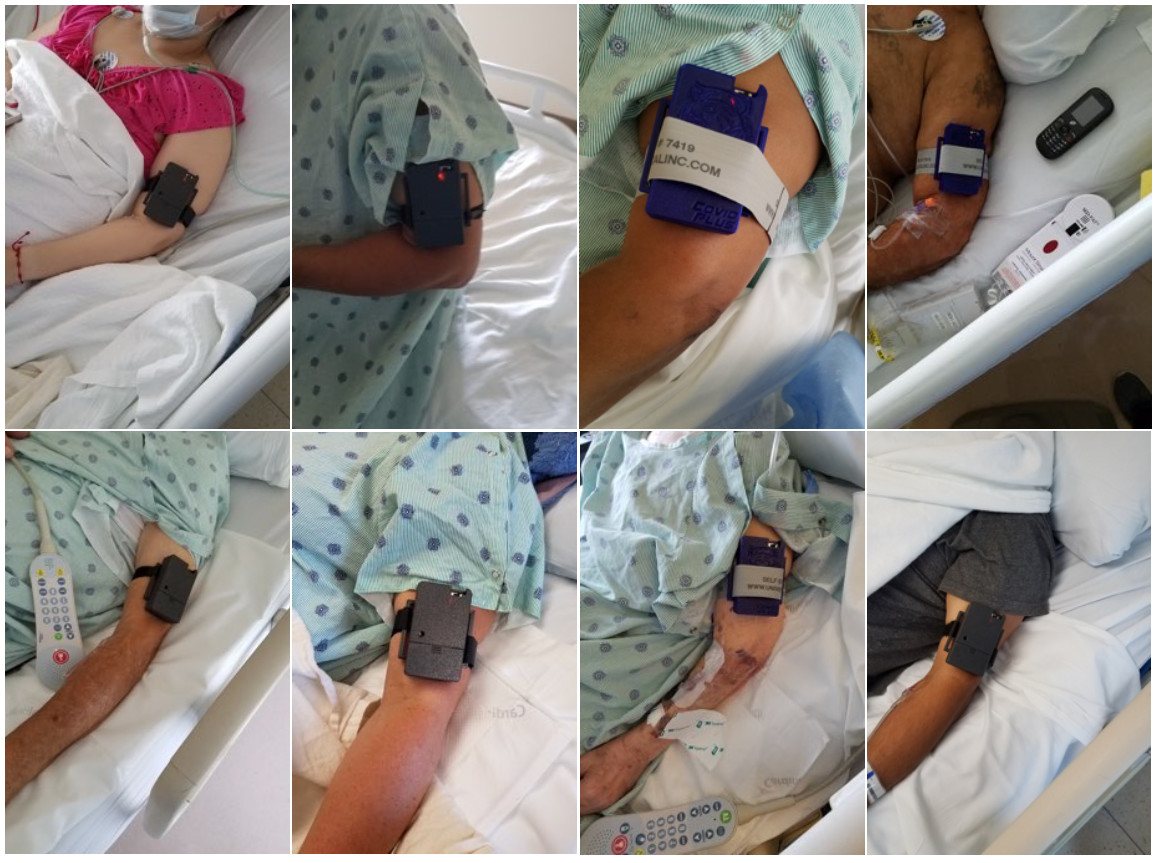
Take the U.S. Food and Drug Administration. Early on, the FDA issued an emergency use authorisation (EUA) to help fast-track clearance for technology that could help the fight against covid-19. That doesn’t mean expedited tech will just magically find a way onto the marketplace. It took until March 19 for the FDA to grant clearance to the Tiger Tech COVID Plus Monitor.
The device itself is a non-diagnostic screening tool worn on the arm that uses light sensors to check for “biomarkers.” Hypercoagulation — or easier-than-normal blood clotting — has been identified as a common abnormality in covid-19 cases. The device itself is meant to be used in conjunction with temperature screening, sort of as a backup method, to limit the spread in public settings like hospitals, schools, offices, theme parks, stadiums, and airports. This tech would be great for reopening, but it’s also not at all clear as to how the Tiger Tech device would be used in the next few months. It’s possible that you never even see one, despite the FDA clearing the device for the market.
Ultimately, these efforts are still worth it, if not for this pandemic, then the next flu outbreak or whatever infectious virus decides to wreak havoc. It’s very possible that the lessons learned from covid-19 can be easily applied to other infectious illnesses. One can only hope.
