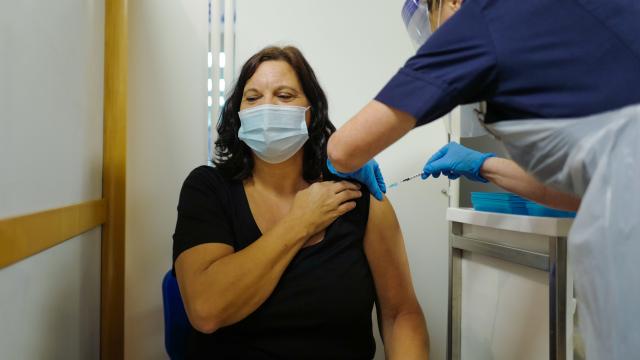
The UK’s top health agency issued a warning on Wednesday that anyone with “significant” allergies to food, medicine, or vaccines should not receive the vaccine, according to a new report from the BBC. The warning comes as two health care workers in England with histories of severe allergies experienced allergic reactions to the new Pfizer/BioNTech vaccine that’s being rolled out this week.
The two health care workers who experienced adverse reactions to the new vaccine were given the shot on Tuesday and both reportedly carry an adrenaline auto-injector (like an EpiPen or similar) to treat their existing allergies in case of emergencies. Other details about their particular allergies and their bad reaction to the Prizer vaccine were not immediately made available.
The UK’s Medicines and Healthcare Products Regulatory Agency (MHRA), roughly Britain’s version of the FDA, did not specifically define what it meant by “significant” allergies when issuing the new guidance on Wednesday.
“As is common with new vaccines, the MHRA have advised on a precautionary basis that people with a significant history of allergic reactions do not receive this vaccination after two people with a history of significant allergic reactions responded adversely,” Professor Stephen Powis, medical director for the National Health Service in the UK, told the BBC.
The two health care workers are reportedly “recovering well,” but details are still scarce. Every office that’s currently dispensing the vaccine in the UK has reportedly been informed on the new guidance for people with allergies. Anyone who receives the vaccine from this point moving forward will be asked about their history of allergies.
Pfizer’s most extensive safety trials showed allergies to the vaccine to the vaccine to be extremely rare, but people with severe allergies were not included in the trials, according to Reuters.
The Pfizer/BioNTech vaccine was approved for emergency use in the UK last week, the first western country to give the green light to a covid-19 vaccine that has completed Phase 3 clinical trials. Thousands of people were vaccinated with the new vaccine on Tuesday in the UK, with health care workers and the elderly the first in line to get the shot.
The UK has been hard hit by the pandemic, with over 1.75 million cases and more than 62,000 deaths this year, according to the latest figures from the Johns Hopkins University online coronavirus tracker. But the U.S. still has the worst coronavirus numbers in the world by far, with over 15.1 million reported cases and more than 286,000 deaths. The U.S. recorded over 213,000 cases and 2,622 deaths on Tuesday alone, according to the Covid Tracking Project, a failure that can be laid at the feet of President Donald Trump and his refusal to adopt a national plan to defeat the virus.
The FDA signalled on Tuesday that all safety and efficacy criteria had been met by the Pfizer/BioNTech vaccine, but the health agency still needs to give its final seal of approval before the shots can be distributed to Americans. FDA officials overseeing the approvals in the U.S. are scheduled to meet again on Thursday.