The Nobel Foundation has awarded scientists John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino the Nobel Prize in Chemistry for their work developing lithium-ion batteries.
Batteries are over a hundred years old, but, pushed by the oil crisis and a search for fossil fuel-free energy resources, researchers at Exxon, including Whittingham, began to develop rechargeable batteries. Thanks to further work by Goodenough and Yoshino, these batteries now appear in most of our portable electronics in some form and are a frequent scientific summary. By 1972, the battery science community came together to discuss how they might incorporate lithium into their devices.
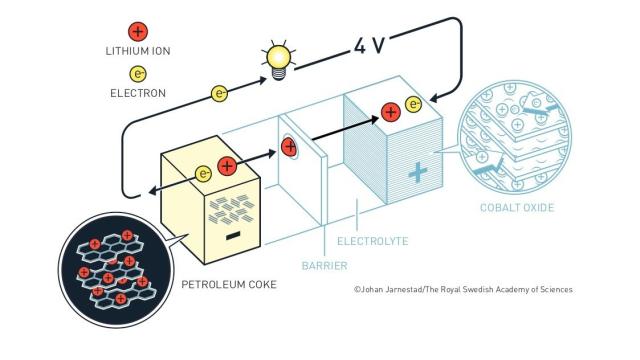
Building on these advances, Whittingham worked to develop a battery with lithium as the electron-supplying anode, but he needed to find an accompanying cathode that would be able to take up all of those electrons and accommodate the lithium ions produced at the anode. He and his collaborator Fred Gamble at Exxon Research and Engineering Company proposed a sort of layered material called titanium disulfide, which could take up lithium ions between its layers. They produced a working rechargeable battery in 1976. Goodenough’s team at Oxford found another material to serve as the cathode that could produce even higher voltages, called cobalt oxide.
But there was a problem. Lithium-ion batteries with pure metallic lithium as their anode would develop whiskers of lithium ions that, if they grew all the way to the cathode, would cause the battery to short circuit. They were also reactive and could explode. Researchers needed to find other, safer materials for the anode as well. Yoshino, working at the Asahi Kasei Corporation in Japan, realised that a material called petroleum coke could be filled with lithium ions could serve as the anode. His group presented a battery based on this anode and Goodenough’s cathode, which overcame all these problems and wouldn’t explode if you dropped something on it.
Like yesterday’s physics Nobel Prize, today’s prize is not without its critics. Plenty of influential battery scientists go unrecognised by today’s prize, and this year’s science Nobels have once again managed to award only men, most of them, white.
Still, the objective impact of this year’s prizewinners is obvious, since researchers have continued improving these batteries since then and billions are now in use across the world. Yes, explosions are still a problem. But, as Steingart pointed out, “I’d argue that there would be no Gizmodo to throw snark were it not for the lithium-ion battery.”
[referenced url=”https://gizmodo.com.au/2018/07/australia-creates-3300-tonnes-of-lithium-ion-battery-waste-a-year-heres-what-we-are-doing-about-it/” thumb=”https://gizmodo.com.au/wp-content/uploads/2018/07/Lithium-Ion-Battery-410×231.jpg” title=”Australia Creates 3,300 Tonnes Of Lithium-Ion Battery Waste A Year: Here’s What We Are Doing About It” excerpt=”A new CSIRO report reveals Australia’s potential to be a world leader in re-using and recycling lithium-ion battery waste.”]