Chinese scientists say they have accomplished something that’s long been a goal in the world of regenerative medicine – giving someone a new, perfectly compatible ear, freshly grown in the lab. What makes the feat a world-first is that the ear was made using that person’s very own cells.
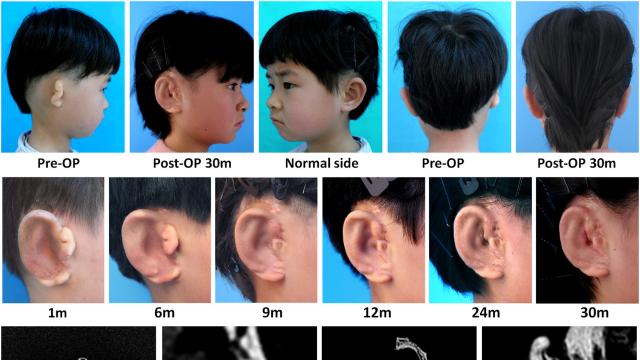
A six-year-old girl, seen above, is the first person to have a new ear successfully made from her own cells. Photo: Cao et al/EBioMedicine
The experimental procedure was performed on five children, ages six to 10 years, with an underdeveloped ear, a condition known as microtia. Currently, the only cosmetic treatments available involve grafting on a synthetic ear, which can be rejected by the body, or an ear roughly sculpted out of rib cartilage by a surgeon, which often looks less than natural.
The researchers created a 3D-printed replica of each child’s normal ear (obtained via a CT scan), but with the dimensions reversed. This replica was then used to create a mould littered with tiny holes and made out of biodegradable material. The mould was filled in with cartilage cells taken from the children’s deformed ear that were further grown in the lab. Over 12 weeks, the cells started to grow into the shape of the mould, replacing bits of it that had already disintegrated. This part-ear/part-mould was then grafted onto the children, some of whom required their own 12-week long procedure that stretched out their skin wide enough to accommodate it.
The first surgery was performed two and a half years ago on a six-year-old, with the latest operation taking place just two months ago. So far, the ears have stayed put, with no signs of the body inadvertently absorbing or rejecting the material. Cartilage has also continued to gradually replace the mould, resulting in a more natural-looking ear over time. The team’s results were published in the journal EBioMedicine.
“It’s a very exciting approach,” Tessa Hadlock, a reconstructive plastic surgeon at the Massachusetts Eye and Ear Infirmary in Boston, told New Scientist, which first reported on this research. “They have shown that it is possible to get close to restoring the ear structure.”
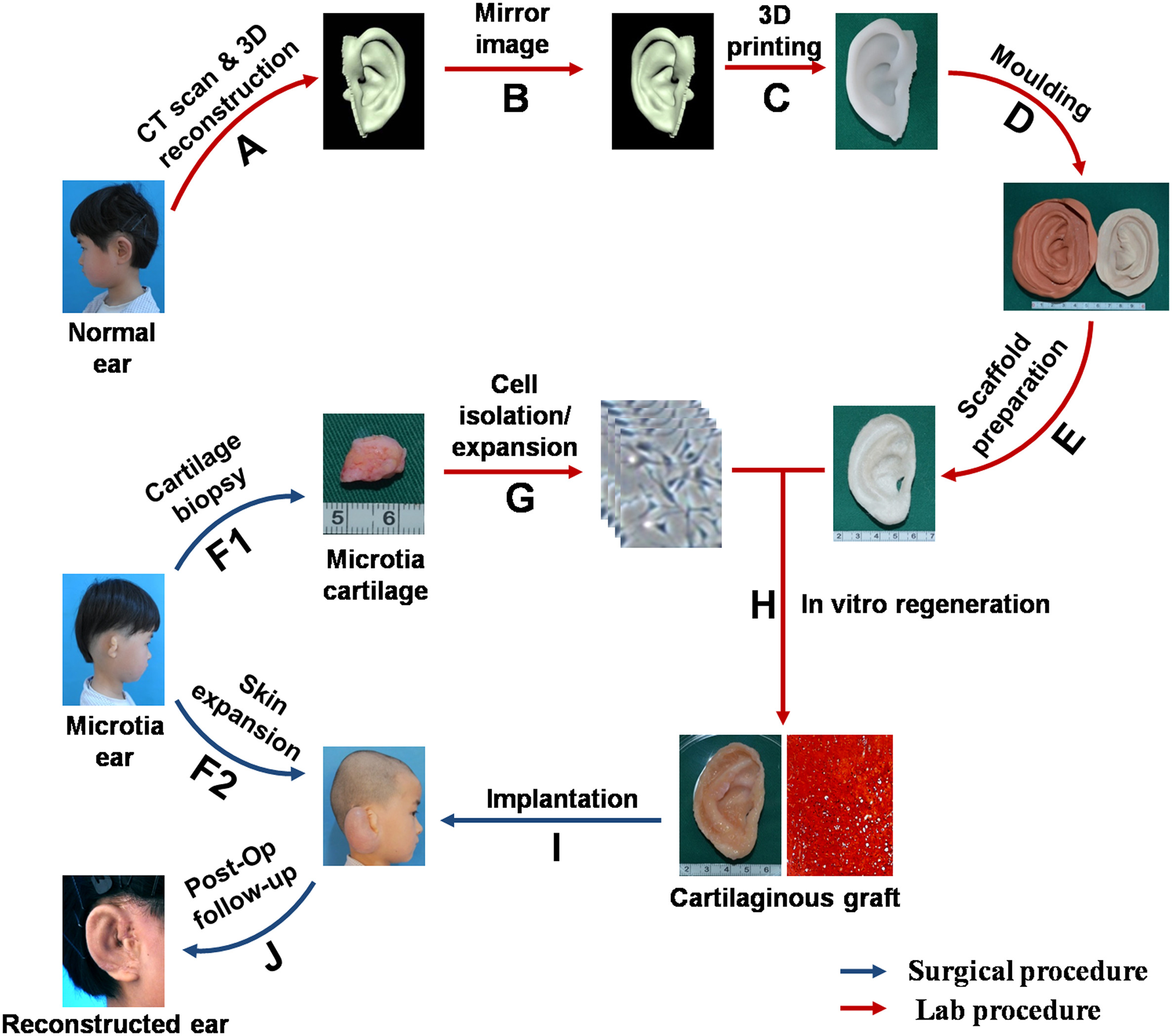
The lengthy process behind the procedure, illustrated above. Image: Cao et al/EBioMedicine
Scientists have been trying to grow the perfect ear replacement for decades. They have been partly inspired by the so-called earmouse: A photograph taken from a 1997 study (which at least one of the current study’s researchers co-authored) that seemingly showed a lab mouse with a human-looking ear growing out of its skin. The ear was actually just cow cartilage shaped to look like one that was grown on a ear-shaped scaffold and grafted onto the mouse, but it nevertheless seeded the idea (and for some people, fear) of someday being able to mass-produce needed body parts and organs.
According to Dr Bruno Peault, a regenerative medicine expert based at the University of Edinburgh in the UK as well as at UCLA, creating a new ear from scratch has also been a tantalising goal because of how simple it would be. Ears are basically just cartilage and skin, so it’d be a stepping stone in learning to bioengineer tissue that can be readily accepted by the body. “It’s a relatively low-hanging fruit,” Peault said, who is unaffiliated with the current study.
Peault and his colleagues in the UK and US have been working on a similar project using 3D-printed scaffolds to grow new ears. Their method, however, uses stem cells taken from the patient’s fat tissue that can be guided into becoming ear cells. Ideally, Peault said, these cells, rather than the cartilage cells used by the Chinese scientists, would make for a more stable base on which to build a new ear from.
“They’re using something that’s not as pure, but may still work,” he said of the Chinese team’s methods.
There is one advantage scientists living in China might have over other researchers like Peault. “In general, things progress faster in countries like China, because the regulations needed to get to clinical trials are much more looser than they are in the US or Europe,” he said. “In the US, it’s a long, difficult, sometimes very frustrating, and expensive [process].”
Peault’s team is still collecting the research needed to win approval for human trials of their method; it might be another one to two years before that might become a reality. Regardless, he is ultimately heartened by the new findings and what they mean for his field. “Certainly there is a bright future for these strategies,” he said.
Meanwhile, the scientists behind the current study plan to monitor their patients for at least five years, in order to make sure the ears stay intact even after the scaffold has completely broken down. They also hope to refine the procedure to avoid the complications seen with some of their patients, such as scarring and a slight deforming of the new ears over time.
[EBioMedicine via New Scientist]