Image: MIT
The current slate of treatments for HIV have been nothing short of life-saving. These antiretroviral therapy (ART) drugs, as they’re known, keep people with HIV healthy and virtually virus-free, with relatively minor side effects — so long as they take the pills daily. The biggest reason why some people can’t keep up with daily treatment is access: Many people, particularly in poorer areas of the world, simply can’t afford a lifetime supply. But there’s also a problem of adherence — as much as 30 per cent of people can’t stick to the needed daily regimen of ART. To help remedy this commitment gap, researchers at the Massachusetts Institute of Technology (MIT) and Harvard Medical School have created a tiny star-shaped cargo container, packed inside a gel capsule, they hope can parse out a week’s supply of ART with one dose.
Their latest research, published Tuesday in Nature Communications, is a “proof-of-principle” animal study. Using pigs, they found their drug delivery system could provide a steady release of three common ART drugs over a week with few drawbacks. “Clinical translation of this strategy would represent a paradigm shift in HIV treatment and prevention,” the authors wrote.
Because some ART drugs are used to prevent HIV infection, via the daily use of pre-exposure prophylaxis (PrEP), they also calculated how much more effective a weekly PrEP pill could be compared to the daily version, based on previous research of how often people take their medications.
“We found that this change would manifest in approximately a 20 per cent increase in efficacy for weekly PrEP as compared to daily PrEP,” senior author Gio Traverso, a research affiliate at MIT’s Koch Institute for Integrative Cancer Research as well as a professor at Harvard University, told me in an email. That could amount to anywhere from 200,000 to 900,000 cases of HIV prevented over the next 20 years in a high-risk part of the world, such as South Africa, depending on how popular PrEP becomes.
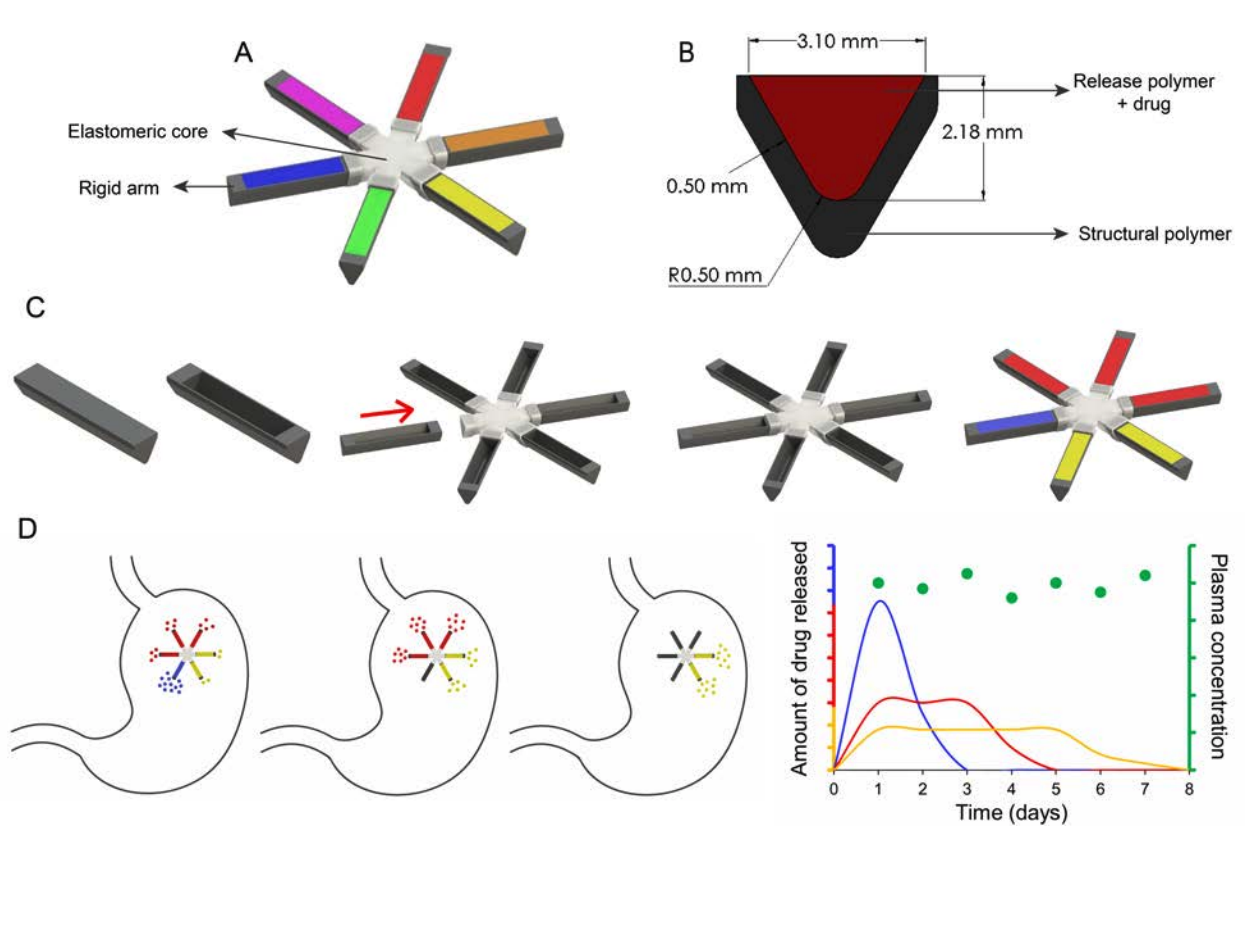
A concept design of how the star-shaped delivery system works to slowly release three different ART drugs over a week’s time in the body. Image: Kirtane, et al/Nature Communications
The delivery system is made out of six rigid “legs” that attach to a central core; each leg is filled with the drug. The legs are coated in a structural polymer that keeps them folded inside the gel capsule and safe being dissolved by stomach acid. Once inside the stomach, the gel capsule dissolves and the legs spring out and unfold into the star shape, making the pill too large to pass down into the small intestine. The inside of each leg is made of a different “drug-release” polymer, meticulously picked out by Traverso and his team to dissolve away at different rates. That ensures each drug payload will release at a different time. After a week, the connections between the legs and the core dissolve away, so the whole thing gets safely flushed out of the body.
This sort of easy customisation ideally means the technology could be a near-universal drug delivery system, not just a platform for HIV treatment. An earlier version of the system was tested with the malaria drug ivermectin, and the team is expecting to develop versions that could work with drugs used to treat tuberculosis, malaria, and several neglected tropical diseases, Traverso said. Each drug would require a different formulation of polymers, depending on the chemical properties of the drug as well as the necessary dosage schedule. Theoretically, Traverso said, the system’s release timeline could be extended to at least a month but possibly more, based on their research. Lyndra, the biomedical company Traverso and others formed around this technology, is planning to focus on these diseases, as well as neuropsychiatric disorders like Alzheimer’s.
As with any promising medical advance, though, the real proof in the pudding will only come through research involving human subjects. To that end, Lyndra is spearheading clinical trials. If everything goes well, Traverso says these trials could be completed within the next year or two.