Image: AP
For the first time, a research team from China used a new technique to fix a blood disorder in human embryos. The scientists performed “chemical surgery” — a procedure that rewrites errors in genetic code instead of snapping and replacing strands of faulty DNA, which is the central strategy employed by the CRISPR gene editing system. The early results look promising, but this technology is still a long way from prime time.
Brazilian Jiu-Jitsu is a fascinating and even unconventional martial art in that its practitioners, rather than coming out all guns-a-blazing with fists, knees, and elbows, use their opponent’s weight and strength against them. This new gene-editing technique, which was introduced last year by a Harvard research team led by David Liu, is a lot like Jiu-Jitsu in terms of its subtler approach. Where CRISPR slashes sloppily through DNA like an unsharpened knife, the “chemical surgery” technique, as Liu calls it, actually goes into the code of DNA itself, flipping misspelled letters, or bases, into their correct form.
A team of researchers from Sun Yat-sen University in China are now the first to use Liu’s invention on an actual human embryo, though the modified embryo was not implanted into a surrogate. As first to use CRISPR on human embryos, though that pioneering attempt to fix abnormal beta-thalassemia genes was met with limited success. By turning to chemical surgery, a technique otherwise known as “base editing,” the researchers were hoping to get superior results.
The new technique is not meant to replace CRISPR, but it could be used to fix inherited disorders caused by single mutations, and in a way that produces fewer unpredictable results. The scientists are claiming limited success, but because the technique sometimes caused new mutations instead of fixing them, proclamations that this is the next big thing in gene-editing should be met with trepidation.
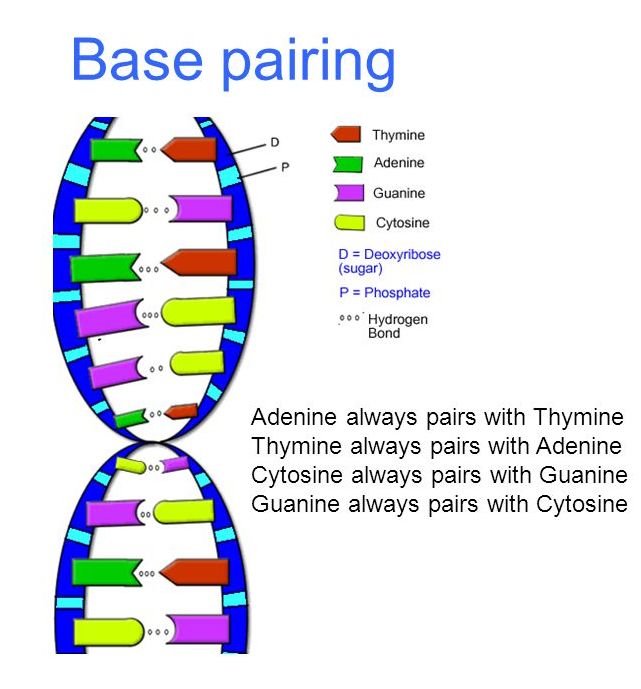
Image: Bio3B
Base editing works by fixing a single error out of the three billion letters that make up our genetic code. The fundamental building blocks of DNA are comprised of four bases: A, C, G, T — letters than correspond to adenine, cytosine, guanine and thymine. The entire set of instructions for building a human are encoded through combinations of these four bases, but every once in a while these combinations get borked. In beta-thalassemia, the disorder is triggered by a lone error to a single base. When this happens, scientists refer to it as a “single point mutation.” These single letter errors account for at least two-thirds of inherited genetic diseases, and because base editing holds great potential for fixing single point mutations, it could conceivably be used to treat an assortment of inherited disorders.
By comparison, CRISPR uses an enzyme (Cas9 nuclease) to remove a chunk of undesired DNA, and then relies on the cell’s response to this double-stranded slice, which it does by making changes in genomic DNA at the target site. But instead of this “cut and paste” approach to gene editing, chemical surgery works by manipulating (i.e. chemically altering) the DNA bases themselves, converting one desired letter to another.
“[Base editing] does not make a double-stranded cut in the target DNA,” explained Liu to Gizmodo. “Instead, base editors directly perform chemical surgery on the target DNA base to convert one base pair to a different base pair (that is, to make a point mutation). Direct chemical surgery is an appropriate analogy because base editors actually catalyze the rearrangement of atoms on the target DNA base to transform that DNA base from a C to something that looks like a T (for example).”
Liu, who wasn’t involved in the new study (aside from the fact that he and his team at Harvard invented the technique), says this approach results in fewer unpredictable results at the target site. In the case of beta-thalassemia, Puping Liang and colleagues turned a G to an A, which corrected the error. To fix the misspelling in the DNA code, the Sun Yat-sen University researchers used an enzyme similar to the one used in CRISPR, but they did so in the manner of a Jiu-Jitsu black belt, rewriting letters instead of coming down with clumsy karate chops.
Liu describes the new paper as “interesting work” that demonstrates base editing’s ability to “directly correct a pathogenic beta-thalassemia mutation in the promoter of the beta-thalassemia gene in cells from beta-thalassemia patients, and in cloned human embryos derived from these patient [skin] cells.” He said the researchers observed “efficient corrections” of the mutation responsible for the blood disorder.
“To my knowledge, the study is the first to use base editing to correct a pathogenic mutation in human embryos,” Liu told Gizmodo, “though some of the same researchers previously published a paper in the same journal showing that base editing could work efficiently in discarded human inviable embryos.”
It’s important to note that base editing can’t fix all mutations, and in its current state of development it can only change a single C to a T or a single G to an A.
“It really isn’t a rival to CRISPR-Cas9. It is a modified version with different attributes,” explained Seth Shipman, a geneticist at Harvard Medical School, in an interview with Gizmodo. “Because only certain base changes are possible with this editor, one challenge has been to find cases where the changes that can be made would potentially fix a disease. Puping Liang and colleagues found one and showed it in this paper.”
Interestingly, Shipman says the Chinese researchers buried the lead in their new paper. Though the technology is intended to treat inherited disorders in germline cells (i.e. sex cells that permanently encode genetic modifications, allowing a person to pass the traits on to the next generation), Shipman says base editing can be used to treat the faulty cells of living persons.
“Their final experiments are in these nuclear transfer embryos as a way of showing whether one might be able to make a germline modification to correct this mutation,” Shipman told Gizmodo. “In the case of beta-thalassemia though, if they were able to edit the…stem cells instead, they may be able to treat or even cure the disease in patients who are currently suffering from it, rather than potentially modify their offspring. That could be a more relevant line of research — and one they hint at in their discussion.”
This is important because somatic modifications are far less controversial than germline tweaks, which become inherited. The downside with somatic modifications, of course, is that each and every person who inherits a genetic disorder has to receive treatment.
Speaking to The Guardian, Robin Lovell-Badge from the Francis Crick Institute in London said the recent experiment “looks very promising,” but cautioned that the modified embryos still had problems. Humans carry a pair of copies in every gene (alleles), he said, and to avoid disease, a sizeable portion of these genes need to be normal. The new approach, said Lovell-Badge, often fixed one mutation in the two pairs, creating “mosaic” embryos, that is, embryos with both healthy and mutated cells.
Importantly, the Sun Yat-sen University researchers aren’t saying the new technique is anywhere near being ready for clinical use, but they say their study “demonstrated the feasibility of curing genetic disease in human…embryos by base editor system.”
Further refinements to the system will now be a central focus as this technology develops, while society contemplates what it all means. It’s currently illegal in many jurisdictions (the US included) for scientists to implant an embryo modified by a gene-editing system — whether it be done by CRISPR, chemical surgery, or any other technique. The era of human genetic modifications is drawing closer with each passing breakthrough, and thankfully the slow rate of progress is allowing society to grapple with the many implications these biotechnologies will bring.