Using stem cells in grown-on 3D scaffolding in a laboratory petri dish, scientists have for the first time created an embryo made entirely from stem cells.
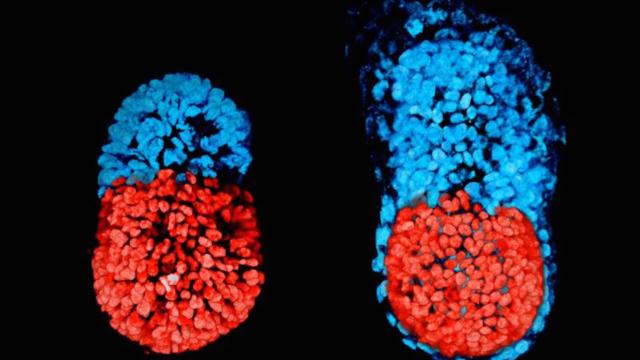
An artificial mouse embryo after 48 hours (right) and 96 hours (left). Image: University of Cambridge
The artificial mouse embryo, detailed this month in the journal Science, is a major step toward creating synthetic embryos that closely resemble natural ones. It could shed light on early development and help improve fertility treatment procedures.
Cambridge biologist Magdalena Zernicka-Goetz, whose work focuses on the development of cell lineages, has long sought the development of an artificial embryo. But without using an egg in some way, scientists have had difficulty getting cells to communicate with each other early in development. Scientists managed to clone Dolly the sheep, for example, without requiring a ram’s sperm, but they still required an egg cell to fuse the cloned adult DNA with.
The Cambridge team got around this issue by taking embryonic stem cells (cells found in embryos that can mature into any type of body tissue) and growing them alongside trophoblast stem cells (the cells that produce the placenta). After growing the two types of cells separately, they combined them in a gel matrix. The two types of cells began to mix and develop together. After four days, the embryos began to resemble normal mouse embryos.
The goal is not necessarily to create a real mouse from these cells — and the science is still a ways from that anyway. Additional types of cells will likely needed to be added to the mix in order for the embryos to actually start developing organs. Even then, the cells may not develop past the very early stages shown in the Science paper.
But being able to study the way the cells develop in the very early days of an embryo’s life could shed important light into early development. The Cambridge researchers, for example, engineered different cell types to glow different colours so that they might track how they behave as the embryo develops. The work provided insight into how those two types of cells work together to form the blue print for the mouse body.
Here’s a video of Zernicka-Goetz explaining the work:
[Science]