An unproven stem cell therapy conducted by a Florida clinic has blinded three patients in an apparent clinical trial gone horribly wrong. The incident showcases the extent to which unscrupulous clinics will take advantage of desperate patients — and how the lack of government oversight contributes to the problem.
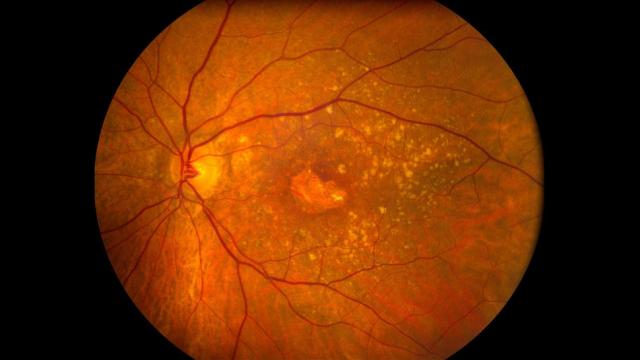
A retina with macular degeneration. (Image: University of Iowa)
As reported in the New England Journal of Medicine, the clinical trial — if it can be called that — involved three women between the ages of 72 and 88 who were suffering from macular degeneration, a common progressive disease of the retina that leads to loss of vision. The women, all of whom were experiencing various degrees of vision loss, sought the help of a Florida clinic, which claimed to be testing a stem cell procedure designed to treat macular degeneration. Sometime in 2015, a week after stem cells were injected into their eyes, the women became blind. Two years later, doctors say there’s virtually no chance the women’s vision will be restored.
The authors of the new report, ophthalmologists Jeffrey Goldberg from Stanford University School of Medicine and Thomas Albini from the University of Miami, said the unfortunate incident serves as “a call to awareness for patients, physicians and regulatory agencies of the risks of this kind of minimally regulated, patient-funded research”.
Stem cells are undifferentiated cells that haven’t quite decided what they want to be when they grow up. Under the right conditions, these immature cells can be transformed into virtually any kind of cell found in the body, which is why they have proven useful in regenerative medicine.
Eventually, scientists hope to be able to use stem cells to regenerate damaged tissue and organs — and possibly even repair the effects of macular degeneration — but we’re not there yet. The only truly effective clinical application of stem cells to date has been in bone marrow transplants, in which stem cells extracted from a donor’s bone marrow are used to produce a fresh blood system for patients suffering from blood disorders such as leukaemia. A recent study showed that there are nearly 600 clinics peddling unproven stem-cell procedures in the United States for a wide range of conditions, including arthritis, autism, cerebral palsy, stroke, muscular dystrophy and cancer.
As noted in the NEJM report, two of the three patients learned about the stem cell trial for macular degeneration on ClinicalTrials.gov, a registry run by the US National Library of Medicine. The listings on this site aren’t fully scrutinised for scientific efficacy. The patients were reportedly under the assumption that they were participating in a bonafide clinical trial, but the consent form and other materials made no mention of a trial. Tellingly, each patient had to pay $US5000 ($6500) for the procedure. This is highly unorthodox for a clinical trial, and it should have been cause for alarm. “I’m not aware of any legitimate research, at least in ophthalmology, that is patient-funded,” Albini said in a statement.
The NEJM study didn’t identify the Florida clinic responsible, but (conveniently) the authors provided the name of the trial: “Study to assess the safety and effects of cells injected intravitreal in dry macular”. A quick Google search calls the trial up, along with the name of the company responsible: Bioheart Inc., otherwise known as US Stem Cell. As the ClinicalTrials.gov page indicates, the study “has been withdrawn prior to enrollment”. According to Goldberg and Albini, the company is no longer performing the procedure, but it is still seeing patients.
The trial itself was a joke, lacking in all the components of a properly designed test. It wasn’t based on prior laboratory experiments, no control group was assigned, no data was collected, and no plans were made for follow-ups.
During the procedure, the patients had some of their fat cells (that is, adipose tissue) removed, along with a standard blood withdrawal. The fat tissues were then processed with an enzyme to draw out stem cells. Once plasma was isolated from the blood and added to the stem cells, the mixture was injected into both eyes of each patient — yes, both eyes. Again, another serious clinical no-no; normally, only one eye would be injected for an experimental procedure like this in the event that something should go wrong. The entire procedure lasted less than an hour.
A week later, all three women were blind. As noted in the NEJM report, the blindness was accompanied by detached retinas and haemorrhaging.
The patients’ severe visual loss after the injection was associated with ocular hypertension, hemorrhagic retinopathy, vitreous hemorrhage, combined traction and rhegmatogenous retinal detachment, and lens dislocation. After one year, the patients’ visual acuity ranged from 20/200 to no light perception.
Goldberg and Albini say the preparation of the stem cells was likely shoddy, and the injections may have been contaminated. Once in the eye, the stem cells could have changed into myofibroblasts, a type of cell associated with scarring.
The Florida clinic, it would appear, was appealing to the desperation of their patients, while taking advantage of a regulatory loophole. As the authors write in their report:
Adipose tissue — derived “stem cells” have been increasingly used by “stem-cell clinics” because of the relative ease of obtaining and preparing these cells. Many of the clinics that provide these stem-cell therapies have done so under the auspices of patient-funded, institutional review board — approved research, and the research is listed on ClinicalTrials.gov without an investigational new drug filing with the FDA.
At the time, the procedure was not subject to FDA approval because the cells weren’t transferred between patients, and because the cells were considered “minimally processed”. The FDA has since revised its requirements, and it now needs approval for these types of procedures. In addition to updating its regulations, the FDA is also clamping down on stem cell clinics.
That’s obviously a good thing, but it’s a little too late for the women involved. This incident shows what happens when regulations and oversight are weak, and how shady companies will take risks with their patients’ health. Certainly food for thought as Trump and his administration start to recreate the FDA in their own image.
We reached out to US Stem Cell Clinic for comment and they responded with this statement:
Founded in 1999, U.S Stem Cell, Inc. has been committed to the research and development of effective cell technologies to treat patients with a variety of diseases and injuries. Since 2001, our clinics have successfully conducted more than 7,000 stem cell procedures with less than 0.01% adverse reactions reported. We are unable to comment further on specific cases due to patient confidentiality or legal confidentiality obligations. Neither US Stem Cell nor US Stem Cell Clinic currently treats eye patients.