Schizophrenia is a complex disease with elusive origins, but the mystery became much clearer today, when a landmark new study based on genetic analysis of nearly 65,000 individuals pinpointed a specific gene and biological process behind it.
The discovery injects new hope into the century-old quest to treat — and perhaps even cure — the debilitating psychiatric disorder. Roughly one per cent of the population suffers from schizophrenia, a disease characterised by hallucinations, emotional withdrawal and a declining cognitive function, beginning in adolescence or early adulthood. Despite decades of research, we’ve made very little progress treating schizophrenia, in part, because it’s been so difficult to nail down the cause.
“Since schizophrenia was first described over a century ago, its underlying biology has been a black box, in part because it has been virtually impossible to model the disorder in cells or animals” said Steven McCarroll, director of genetics at Stanley Center for Psychiatric Research. “The human genome is providing a powerful new way in to this disease.”
McCarroll would know, too. In 2014, he participated in a massive international collaboration which pinpointed more than 100 regions of the human genome carrying risk factors for schizophrenia. Now, in a paper that appears in Nature, McCarroll and his colleagues have revealed a specific gene and biological process underlying the strongest risk factor of all.
On the surface, the culprit behind schizophrenia sounds a bit odd. It’s a variant in the major histocompatibility complex (MHC) — a set of proteins that decorate the surface of your cells — that binds to foreign molecules and presents them to the immune system. But McCarroll’s new study, which looked at the DNA of nearly 29,000 individuals with schizophrenia and 36,000 without, showed that this particular MHC variant causes the expression of a gene known as C4 to go into overdrive.
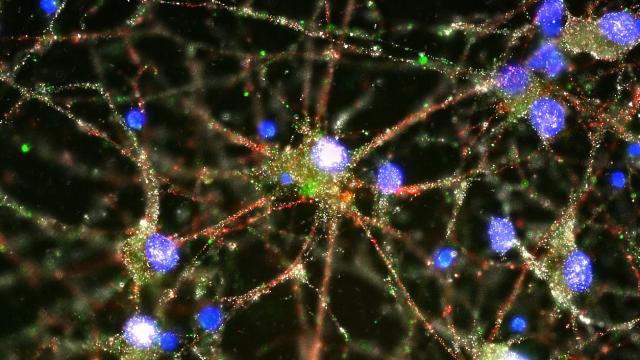
And it so happens that C4 is present at neuronal synapses, the connections between neurons that transfer chemical and electrical signals in your brain. On a cellular level, too much C4 can reduce the number of synaptic connections, a process known as “synaptic pruning”. On a human-scale, this can lead to schizophrenia.
The findings represent a major breakthrough for neuroscience, but McCarroll and his colleagues see their discovery as the first step on a path toward new and more effective treatments.
“Because the molecular origins of psychiatric diseases are little-understood, efforts by pharmaceutical companies to pursue new therapeutics are few and far between,” said Bruce Cuthbert, acting director of the National Institute of Mental Health “This study changes the game.”
[Nature]
Top: Imaging studies showed C4 (in green) located at the synapses of primary human neurons, via Heather de Rivera / McCarroll lab.