Researchers in China are reportedly the first to use a powerful gene editing tool to produce super-muscled dogs. The goal is to create test subjects that mimic degenerative human diseases, but the breakthrough also raises the prospect of customised pets.
A cheap and powerful gene editing technique called CRISPR is taking the science world by storm. Over the past year, biologists have used the genetic cut-and-paste tool to genetically modify human embryos, produce supersized fruit, and create double-muscled pigs and micropigs. Other animals involved in CRISPR work include goats, rabbits, and monkeys.

Image at left: Hercules and Tiangou, the world’s first gene-edited dogs. They may not look particularly extra-muscled, but the researchers say Tiangou (the one of the right), features larger thigh muscles compared to her littermates, along with other “obvious muscular phenotype.” (Journal of Molecular Cell Biology via Technology Review)
Now, as Technology Review reports, scientists in China have used CRISPR to knock out the myostatin gene (MSTN) in beagles, which roughly doubles their muscle mass. The details of this work can now be found at the Journal of Molecular Cell Biology.
The researchers say that the super muscly beagles could be used for hunting, police, and military applications, but the ultimate purpose of the experiment is to create dogs with other DNA mutations, including those similar to Parkinson’s and muscular dystrophy.
“The goal of the research is to explore an approach to the generation of new disease dog models for biomedical research,” noted study co-author Liangxue Lai in Technology Review. “Dogs are very close to humans in terms of metabolic, physiological, and anatomical characteristics.” Lai is a researcher with the Key Laboratory of Regenerative Biology at the Guangzhou Institutes of Biomedicine and Health.
TR writer Antonio Regaldo describes the experiment:
In the end, of 65 embryos they edited, 27 puppies were born, but only two, a female and a male, had disruptions in both copies of the myostatin gene. They named the female Tiangou, after the “heaven dog” in Chinese myth. They named the male Hercules.
Lai and his colleagues reported that in Hercules, the gene editing was incomplete, and that a percentage of the dog’s muscle cells were still producing myostatin. But in Tiangou, the disruption of myostatin was complete and the beagle “displayed obvious muscular phenotype,” or characteristics. For example, her thigh muscles were large compared to those of her litter mates.
The beagles are similar to super-muscled Belgian Blue cattle and whippet dogs, both of which have been bred in such a way that they now “naturally” suppress myostatin production at the genetic level.
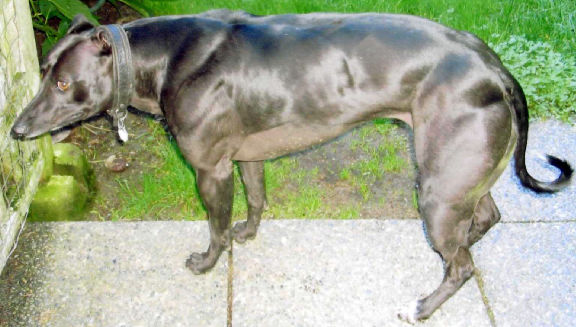
A whippet with the myostatin mutation (PLOS/CC)
When the myostatin gene is disrupted, the body fails to produce the muscle-inhibiting protein manufactured by the gene. The end result is extra muscle mass. Some humans have actually been born with this mutation.
By studying this genetic quirk, and related conditions involving muscle buildup and decay, scientists hope to treat degenerative disorders in humans. As it stands, however, it is currently illegal and grossly premature to use CRISPR on human subjects. But as this research shows, human applications may not be too far off in the distance.
The researchers say they have no plans to breed the jacked-up beagles as pets, but it’s obvious that this is where the technology is headed. In future, dogs and other animal companions could feature an assortment of genetic manipulations, including changes to intelligence, temperament, physical proportions, and colour patterns.
Much more at Technology Review. You can find the entire scientific study at the Journal of Molecular Cell Biology: “Generation of gene-target dogs using CRISPR/Cas9 system“.
