Funky, floral, complex. No, this is not a description of a piece of vintage wallpaper. These are some of the words that are used to describe the enormous variety that exists within the world of beer. Whether you are enjoying the outdoors on a sunny day or sitting by the fire on a cold, winter night, there is a beer to match every occasion. In honour of this tasty drink, we have put together a compilation of PLOS ONE articles dedicated to the study of beer and the yeast and bacteria that mould its complexity.
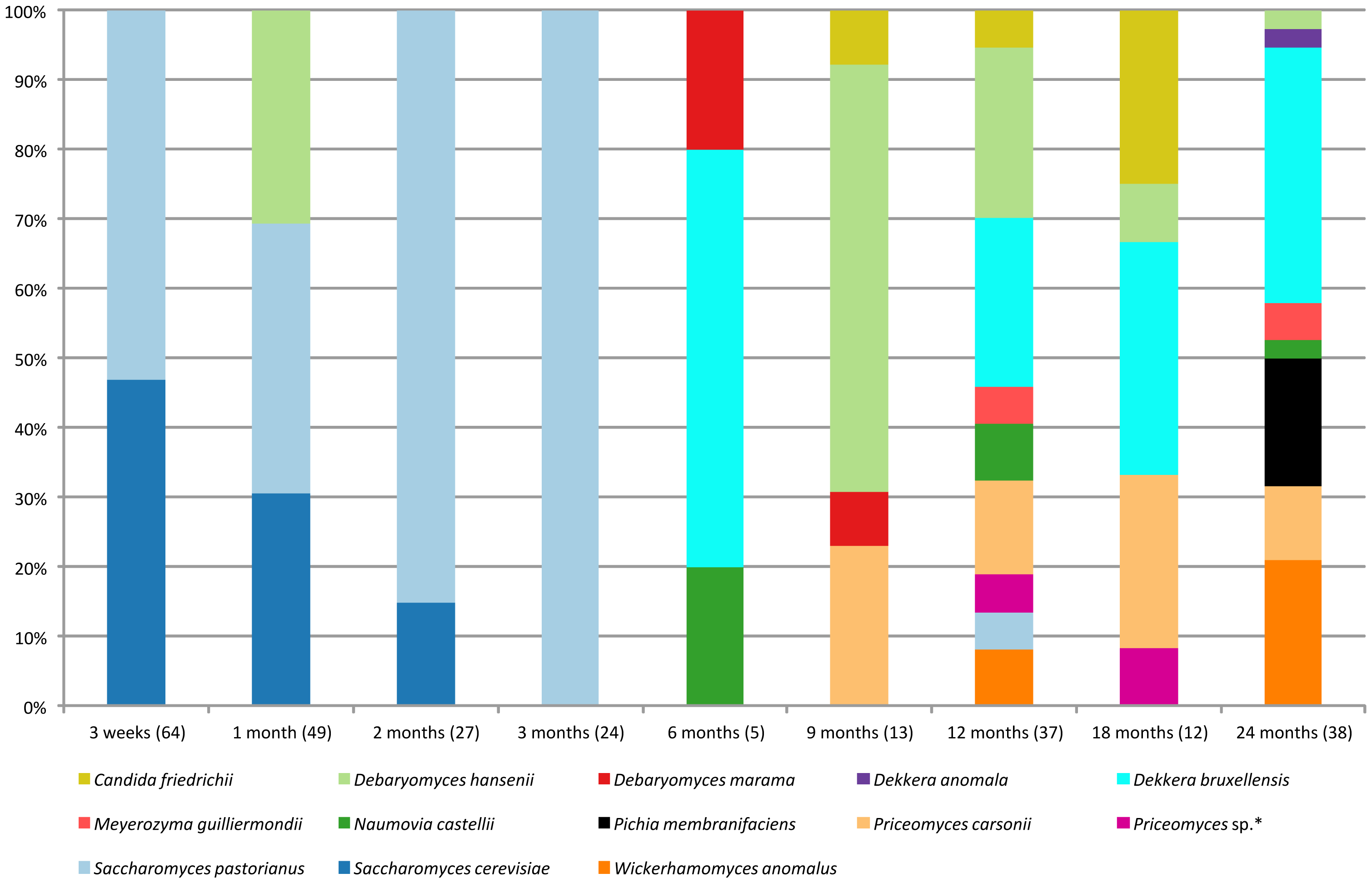
Figure 4 of the published article shows the different isolates found over time in the DYPAI and UBAGI agars of batch 1.
The brewing process for most beers includes heating hops and grain in nearly boiling water, before cooling the liquid down and adding a carefully selected yeast strain. Lambic sours, on the other hand, undergo a process known as spontaneous fermentation. Contrary to its name, spontaneous fermentation is a lengthy and controlled process that lasts, on average, a couple of years. The process itself depends on the strains of ‘wild’ bacteria and yeast that are already present in the environment to produce a tart flavour; normally, the presence of wild microbes causes a type of contamination referred to as ‘infection.’ ‘Infected‘ beers are most easily identified by their undesirable taste, colour, and smell. Therefore, as you can imagine, the microbiome of beer and how it may change during the fermentation process are of much interest to researchers and beer enthusiasts alike.
By looking at the types of microbes that colonised beer samples collected from the Belgian brewery Cantillon, prized by beer-lovers around the world, the authors of one PLOS ONE study were able to see a successive pattern in the beer’s microbiome during the fermentation process. Samples were taken from two batches that were started a month apart and cooled at different temperatures, to see how the microbial biomes varied.
The image above shows the microbial composition over time from isolates in some of the agars of batch 1. Despite some initial variety in the types of identified microbes, and a considerably high degree of species diversity overall, both batches had similar progressions in microbial content as well as a similar microbial composition at the end of fermentation; they consisted of mainly Pediococcus damnosus — a species of gram-positive bacteria that frequently grows in wine and beer. In this case, it may be that spontaneity and different starting points could all lead to a similar microbial ‘colony.’
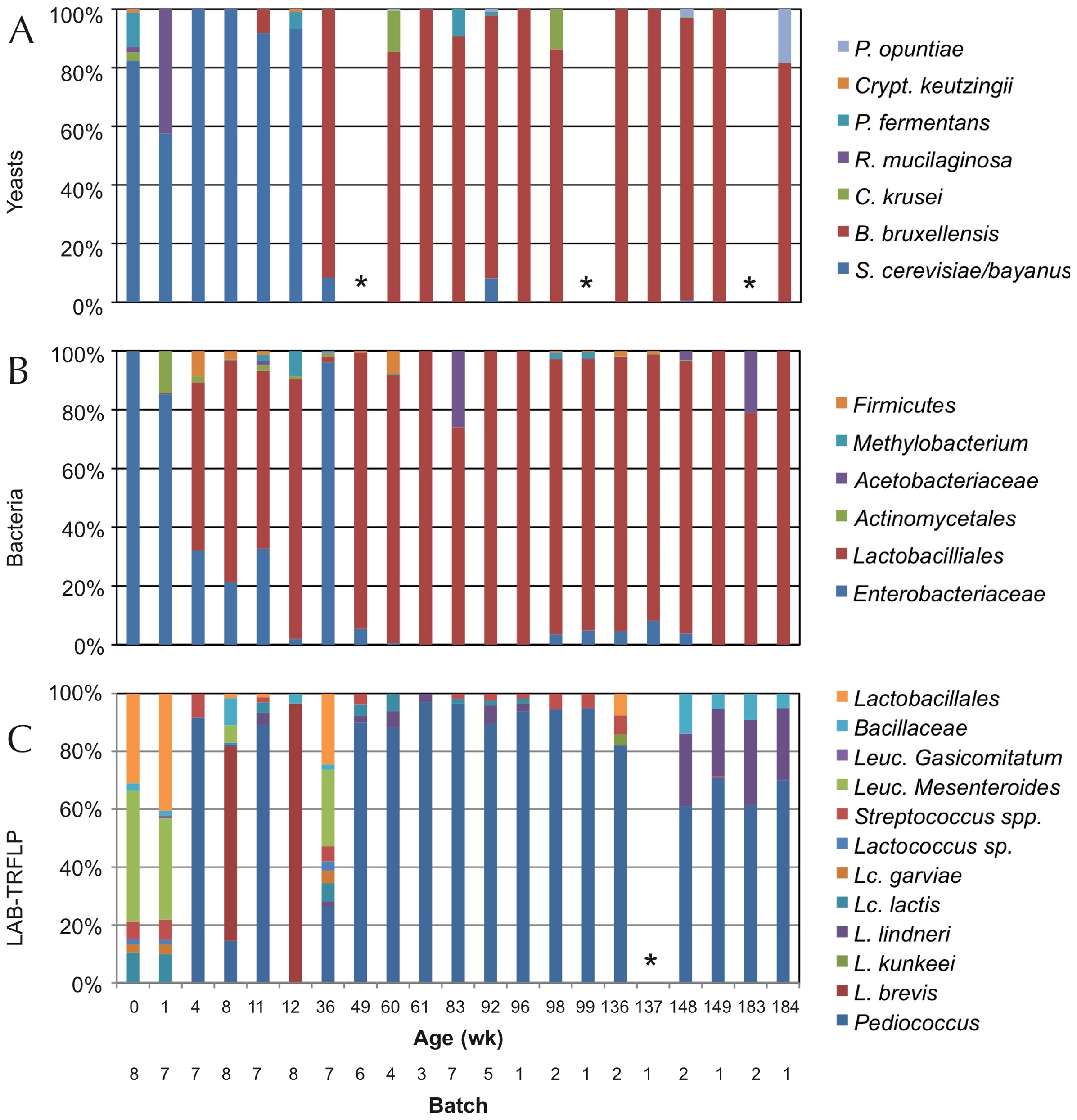
Figure 2 of the published article shows some of the species found in the different batches of ACA over a three year period. Panel A shows the yeast, Panel B is the bacteria, and Panel C shows the lactic acid bacteria.
The American coolship ale is a type of beer that also utilises the power of wild yeast and spontaneous fermentation, and is modelled after the above-mentioned Lambic style. In a 2010 PLOS ONE study, researchers investigated the microbial profiles of multiple batches of American coolship ale from a single brewer in the Northeastern United States, to see if they could establish a “microbial baseline” for this type of beer. The authors collected samples from 8 different batches throughout the 3.5- year fermentation process, and found that while the yeast and bacterial content of the beer started off with a diverse number of species, it ultimately shifted to being composed primarily of B. bruxellensis. B. bruxellensis, more commonly known as Brettanomyces bruxellensis, is the type of yeast responsible for giving beer a distinctly ‘funky,’ lightly tart flavour — it’s so distinct, in fact, that its characteristics are commonly described as ‘Bretty.’
The authors describe this particular microbial succession as likely being caused by the constantly changing environment of the beer. The strains of bacteria and yeast that initially colonised the beer produced carboxylic acid, which can limit the growth of other microbes. Once these early microbial inhabitants died off, Saccharomyces, a type of yeast commonly used in food production, and Lactobacillales were then afforded limited competition and could jump in for the main fermentation process. In the image above, the authors show how the yeast and bacterial profiles changed over time for each of the batches. They explain that since the microbial profiles and their progression are similar across all of the batches, this could be evidence that there are resident brewhouse microbiota that take over during fermentation.
It’s worth noting that studies conducted prior to this one have shown that the microbial profiles of Lambics also ultimately end up being primarily composed of B. bruxellensis, though the smaller communities of microbes differ from those found in American coolship ales.
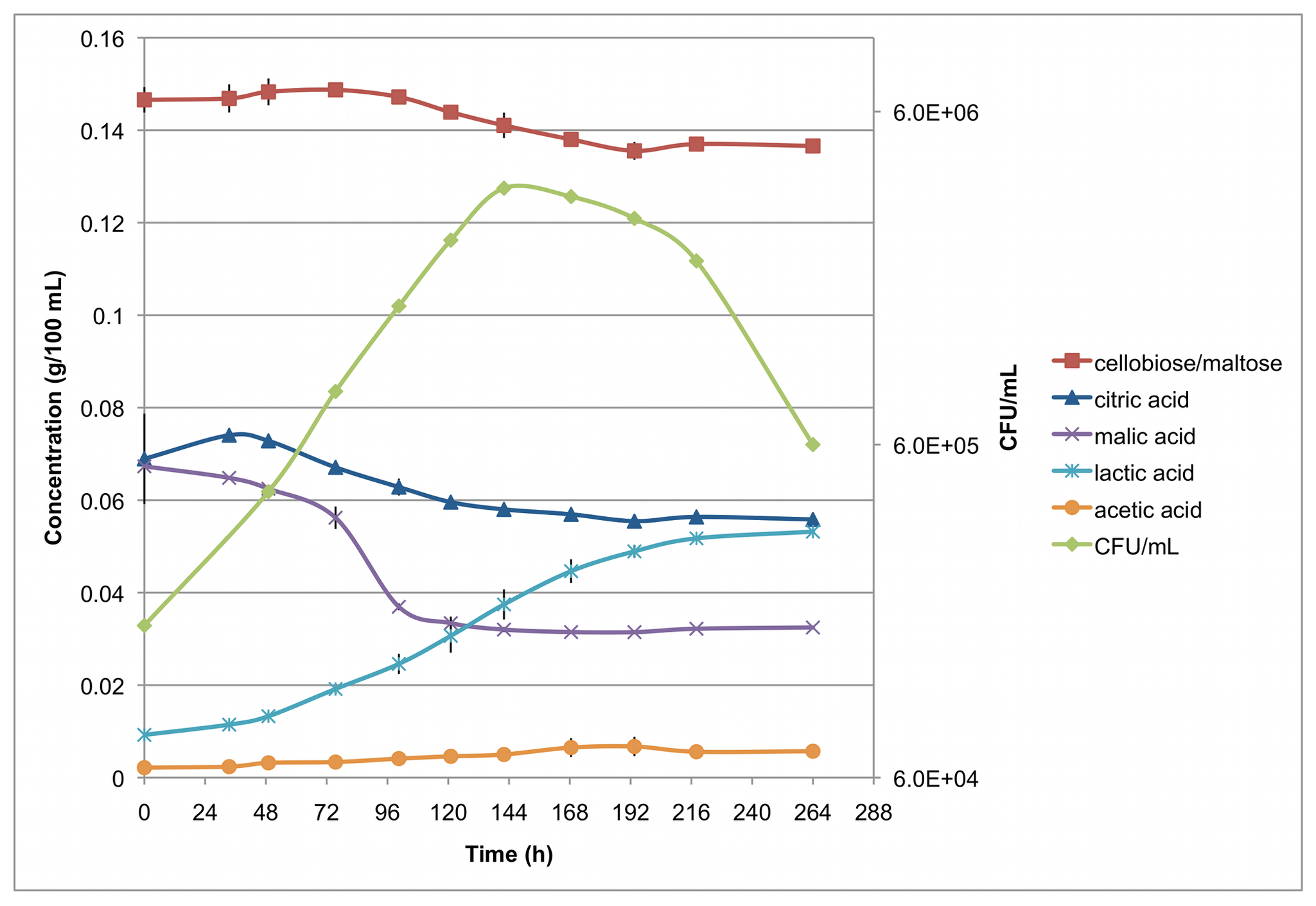
Figure 4 of the published article shows how the bacterial content changed in different concentrations of various acids and maltose over time.
While some beers, such as the previously described sours, thrive with exposure to naturally occurring microbes, others can be ruined by it. During a ‘normal’ brewing process, it is important to ensure that all equipment coming in contact with the beer has been sterilised so that contamination or infection can be avoided. The lactic acid bacteria that helps sours achieve qualities such as their distinctive aroma, may cause other beers to spoil. Luckily, drinking a spoiled beer does not put you at a huge risk for getting sick; they are generally just unpleasant tasting and not very drinkable. Many types of bacteria are unable to grow amid hops, ethanol, and a highly acidic environment; however, a few species have grown to overcome these obstacles.
To better understand the mechanisms employed by these bacteria, researchers of this PLOS ONE study conducted a type of next-generation sequencing called transcriptome sequencing on one of the culprits of beer spoilage: a strain of Gram-positive bacteria called Pediococcus claussenii. The above image shows how the bacterial levels changed over time in relation to the concentrations of various acids present in the beer. Using transcriptome sequencing allowed these authors to determine which genes are used by bacteria when they grow in acidic, low-nutrient environments. While many of these mechanisms are still not well understood, the authors identified genes that may play a key role in the bacteria’s adapted ability to live in these conditions, such as a modification of the cell membrane to resist the acidic environment. Developing a better understanding of how these bacteria are able to live in beer may help avoid contamination in the future.
While beer is enjoyed by many, most don’t give much thought to the science behind the craft. As indicated here, even spontaneous fermentation is a carefully conducted and complicated process that has evolved greatly over the last 7,000 years.
There is a special place where scientist and beer lover unite, as shown by the research articles presented above, and more open access research could mean the potential for better beer, so cheers to all the beer geeks out there!
Citations:
Spitaels F, Wieme AD, Janssens M, Aerts M, Daniel H-M, Van Landschoot A, et al. (2014) The Microbial Diversity of Traditional Spontaneously Fermented Lambic Beer. PLoS ONE 9(4): e95384. doi:10.1371/journal.pone.0095384
Bokulich NA, Bamforth CW, Mills DA (2012) Brewhouse-Resident Microbiota Are Responsible for Multi-Stage Fermentation of American Coolship Ale. PLoS ONE 7(4): e35507. doi:10.1371/journal.pone.0035507
Pittet V, Phister TG, Ziola B (2013) Transcriptome Sequence and Plasmid Copy Number Analysis of the Brewery Isolate Pediococcus claussenii ATCC BAA-344Tduring Growth in Beer. PLoS ONE 8(9): e73627. doi:10.1371/journal.pone.0073627
This article first appeared on PLOS Blogs and is republished here under Creative Commons licence. Top image by Crystal Luxmore under Creative Commons licence.
