With Apple Watch quickly approaching its April release month, app developers are giving us a better sense of the wearable’s capabilities than the designers at Cupertino. For instance, we know how the watch will work with your car or draw up a to do list. Now its health merits are getting some attention.
With help from DexCom, a company that makes monitors for diabetics, the Apple Watch will be one of the first wearables to bring glucose tracking to your wrist. The Apple Watch itself will only act as a display for the information being pumped out every five minutes by DexCom’s continuous glucose monitor or CGM, a hair-thin sensor embedded under the skin.
DexCom teased its intentions to bring glucose monitoring to the Apple Watch at CES 2015, as pictured below with the company’s current iPhone app. Diatribe, who originally reported the CES news, also says that DexCom’s new CGM will also integrate with Apple’s Healthkit platform, which until now has largely been associated with fitness apps. Doctors may also benefit from the partnership as Apple’s Healthkit adoption rate in hospitals is outpacing Google Fit and Samsung’s S Health.
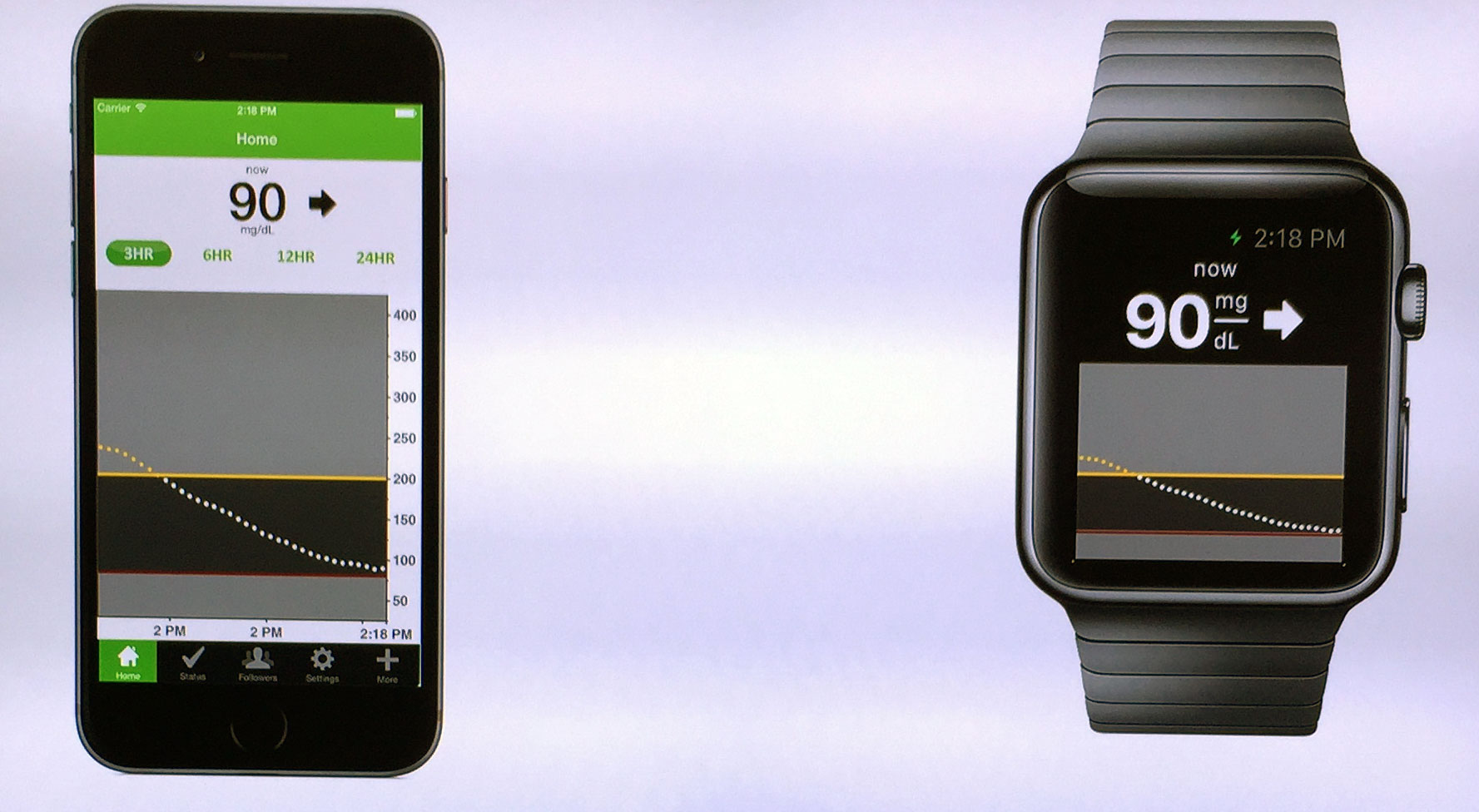
The Apple Watch isn’t the first smartwatch to bring CGM monitoring to a wrist wearable. The Nightscout CGM system, which is an open source project that allows remote access to DexCom sensors, has already developed an app for Pebble. It’s been hoped since the Apple Watch was announced that the wearable would follow in Pebble’s footsteps, and that seems to be what’s happening.
Last summer Reuters reported that Samsung, Apple, and Google are all investigating how to incorporate glucose monitoring into wearables. The one hurdle being that any device marketed for diabetics would fall under the Food and Drug Administrations stringent regulations on Class III medical devices. The CGM sensor in this case is still considered a Class III device, but because of a recent rule change, any apps or software associated with the wearable device only needs to be registered with the FDA, meaning DexCom’s Apple Watch app can bypass the agency’s tortoise-speed approval process and be ready to go when the wearable ships in April, according to The Wall Street Journal.
So it seems the FDA’s relaxed regulations on diabetes software has given developers some room to work, and they’re taking advantage. [The Wall Street Journal]
