Since graphite — the dark material used in regular old pencils — and diamonds are both made from carbon, it’s technically feasible to turn the former into the latter. You just need to apply a little pressure — about 150,000 times what the atmosphere on Earth’s surface is like. But researchers at Stanford University claim to have found a shortcut.
After all, not everyone has access to machinery capable of generating that much pressure — but that might no longer be needed. The researchers stacked four incredibly thin sheets of graphite — microscopically small — onto a platinum metal support structure, and then topped it all of with a bit of hydrogen.
The Stanford team was actually trying to create a superior alternative to silicon, but instead they discovered the addition of hydrogen started a domino-like chain reaction that altered the structure of the graphite layers. Instead of being soft and brittle — which is what makes it ideal for pencils — the graphite became strong and sturdy like diamond.
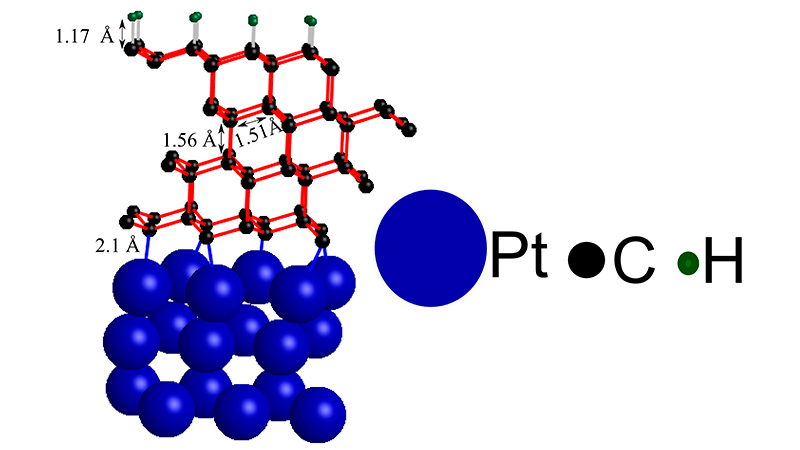
But while the restructured graphite behaves like diamonds, it’s not going to replace the oversized rocks used in engagement rings and other jewelry. The diamonds produced using this method are more suited for industrial applications like coatings that make cutting blades sharper and more durable, or electrochemical sensors. And since the process is much easier, producing diamonds this way should be considerably cheaper as well. [Stanford SLAC via Gizmag]
