The last time Earth’s oceans were this acidic, a six mile-wide sulphur-rich space rock had just smashed into the Yucatan Peninsula, unleashing a deluge of acid rain that exterminated all sea life in the the top 400 meters of the water column. Now, some 65 million years after the Cretaceous extinction, human activity is threatening to similarly decimate the ocean’s ecosystem — this time, from the bottom up.
How the Oceans Went Out of Whack
Under natural conditions, carbon dioxide is continuously transferred between the ocean, atmosphere, and continents in a delicately balanced process known as the carbon cycle. CO2 is pulled from the atmosphere by photosynthetic plants, which form the base of both terrestrial and oceanic food webs. It’ then subsequently sequestered in sediment when those plants — as well as the animals that feed on them — die and decompose. It’s a nice trick and it helps keep us all breathing.
Simultaneously, a roughly equivalent amount of carbon enters the atmosphere due to air-sea gas exchanges, as well as the respiration of sedimentary microbes as they decompose dead organic matter. Along with the nitrogen and water cycles, this carbon cycle is one of the primary facilitators of life on Earth, constantly recycling the limited supply of carbon that forms the base of every organism alive today.
But since the dawn of the industrial revolution, human activity — specifically, burning coal to produce energy — has upended the balance of the carbon cycle. The concentration of CO2 in the atmosphere has jumped from 280 ppm prior to industrialisation to nearly 400 ppm today. We’re pouring more CO2 into the atmosphere than the system can sequester. This excess of atmospheric greenhouse gas has not only resulted in global warming but wreaked havoc on the ocean’s chemistry as well.
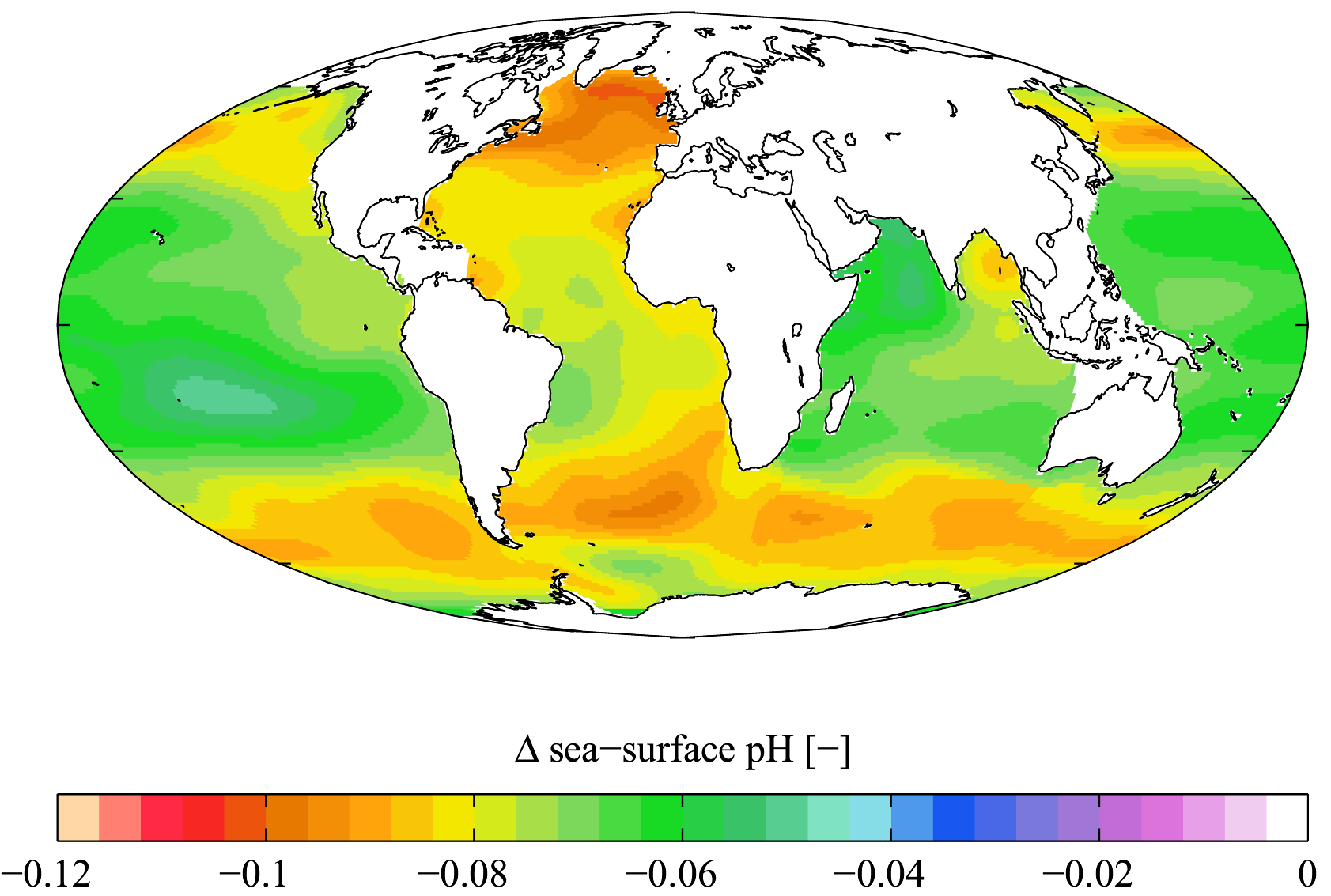
When carbon dioxide enters the ocean, it reacts with seawater to create carbonic acid. This acid in turn produces a secondary reaction, splitting into separate bicarbonate and hydronium ions, which lower the water’s pH level. The more CO2 present in the atmosphere, the more gets absorbed by the oceans, and the lower the water’s pH will become.
Current scientific estimates suggest that the oceans are absorbing roughly 25 per cent of the CO2 we produce each year, with another 45 per cent remaining trapped in the atmosphere, and the rest being absorbed by terrestrial plants. Between 1751 and 1994, the surface ocean pH has dropped from an estimated 8.25 to 8.14. That may not seem like much but remember pH is logarithmic, just like the Richter Scale, so a .11 decrease constitutes a 30 per cent increase in acidity. And if acidification rates continue at their present pace, the pH of the world’s oceans could drop another .5 units — roughly triple the acidity they are right now — by 2100. This would be cataclysmic for sea life and humanity alike.
What This Means for Sea Life

While an added abundance of atmospheric C02 may be a boon to plant life, the resulting acidification it causes is seriously impairing the development of oceanic calcifying organisms — everything that lives in a calcium-based shell from the phytoplankton, zooplankton, and corals that form the base of the food web to mollusks and crustaceans like clams, oysters, crabs, and lobsters.
Normally, there’s a supersaturation of carbonate ions, which these animals process into aragonite for use in their shells. However, as the pH decreases, calcium carbonate becomes more soluble which reduces the concentration of available carbonate ions. And not only does this reduce the rate at which organisms can build their protective structures, it also increases the rate at which existing shells dissolve. They’re literally being melted away by increasingly corrosive seawater.

And it’s not just shellfish that are at risk. Decreased pH levels have been linked to a number of other adverse effects — both direct and indirect — such as the CO2-induced acidification of body fluids, known as hypercapnia, the reduced metabolism in jumbo squid, slowed embryonic development in Atlantic longfin squid, the inability of juvenile clownfish (poor Nemo!) to hear and smell approaching predators, and the diminished echolocation capacity of dolphins and whales.
Nowhere, though, is the effect more clearly illustrated than in coral. Both tropical and deep sea coral species, whose calcium carbonate homes form reefs that support entire ecosystems — acting as both nurseries for a number of commercial fish stocks as well as habitat for countless other species — are showing slower rates of growth than in the past and are suffering from the effects of coral bleaching at unprecedented levels. In 2005, for example, nearly half of the coral around the Virgin Islands and Puerto Rico were lost in a single year to mass bleaching events.

Image: Acropora
The loss of their coral homes only serves to amplify the pressure exerted on existing fish and crustacean populations from overfishing, habitat loss, pollution, and rising sea temperatures. It won’t be long before humanity is directly affected too.
What This Means for Humans
The ocean acts as the primary source of protein for over a billion people worldwide. The US commercial fishing industry exported more than $US5.1 billion of fish products in 2012 alone while providing employment for more than a million Americans. We are the fifth-largest seafood producer behind China, Peru, India, and Indonesia — catching just 3.8 per cent of the global total annually.
Ocean acidification threatens to topple this industry in the near term if steps are not taken to correct it. The populations of popular shellfish like lobsters, crab, scallops, shrimp, oysters, mussels, and clams are in danger of collapse as the concentration of carbonate ions continues to decline. What’s more, the increased water acidity is doing strange things to crab stocks.

Image: Sasha Isachenko
Alaskan Red King Crabs — the centrepiece of the Alaskan crabbing industry, which fetched $US92.5 million for just 14.8 million pounds in 2011 — show a 100 per cent increase in larval mortality (twice as many die) when raised in acidified water, though the less sought-after dungeness crabs, which live in the same areas as King Reds, are less unaffected by the pH change. Maryland Blue crabs, on the other hand, will grow three times their average size when raised in lower pH waters and become extremely aggressive predators. Still, should these populations collapse, the damage to the regional fishing industry — not to mention the prices at your supermarket — will take decades to repair.
What We Can Do About It?
Since ocean acidification (like global warming) is the result of human activity, it therefore can be mitigated by changing the way we interact with the environment.
One obvious answer is to simply reduce the amount of CO2 we’re discharging into the air, though that is far easier said than done. While the world’s governments continue t0 work towards a political solution (see: the Kyoto Protocol) and coastal fisheries simultaneously strive to both slow the rate of acidification and adapt to changing water chemistry, there are a number of steps individuals can take to reduce their carbon footprint. And while reducing your personal carbon emissions may not make a very big impact, the actions of 6 billion individuals taken together could very well save the world. [PhysOrg – Wiki 1, 2, 3 – NOAA 1, 2, 3, 4 – NRDC – WHOI – EPA – Seattle Times – Real Science]
top image: Ethan Daniels
