The medical profession has long dreamed of an ideal delivery system for getting drugs to wherever in the body they’re needed most. Nanoscientists at the University of San Diego have come up with a novel means of doing so: why not fire the drugs at the intended targets, using tiny little cannons?
Early in the 20th century, a German physician named Paul Ehrlich came up with the idea of focusing the delivery of just the right dose of a therapeutic drug at a specific target, rather than waiting for it to gradually work its way through the body via the bloodstream. This gets more of the drugs to where they’re needed most, much more quickly, with far fewer nasty side effects. He called it a “magic bullet”.
Joseph Wang and Sadik Esener, both nano engineers at UCSD, took the notion of a magic bullet literally when they decided to build microcannons that exploit ultrasound to shoot teensy bullets containing therapeutic agents deep into diseased tissue. They described their work in a recent paper in the journal ACS Nano.
We tend to associate ultrasound primarily with medical imaging applications, but it has long been of interest to researchers for various therapeutic uses. For instance, its high-frequency sound waves can loosen stubborn plaque on teeth (it’s now a common tool in dentistry) and staunch bleeding, particularly in the tiny vessels found in vital organs. And it’s increasingly of interest for targeted drug delivery.
Ultrasound is useful for this type of thing because it is largely non-invasive, and the ultrasonic waves can be focused on specific targets, such as a cancerous tumour. The technique not only triggers the release of drugs encased in small delivery packages known as micelles — via sheer stress and shock waves generated by bubbles (known as cavitation) — it also helps the drug permeate deeper into the tissue by making the cell membranes slightly more porous.
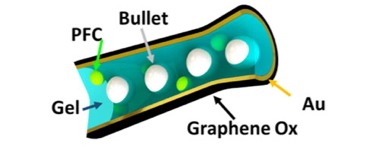
The first step for Wang and Esener was to build the microcannon — no small feat, given that it measures 5 micrometres, or about the thickness of spider silk. The UCSD scientists did this by punching holes in a membrane, like skin with pores, and then spraying the exterior of the holes with a coating of graphene oxide, followed by a second coating of gold.
Then they had to “load” the cannon with 1-micrometre nanobullets (about the size of the HIV virus) made of silica and encased in a liquid gel. That gel also contained a perfluorocarbon (PFC) as a propellant — because without a propellant, how do you shoot the cannon? PFC starts to vaporise when you blast it with an ultrasonic pulse, and this produces teensy gas bubbles that expand rapidly. It’s that rapid expansion that “fires’ the nanobullets out of the microcannon. Without the PFC, the microcannon just won’t fire.
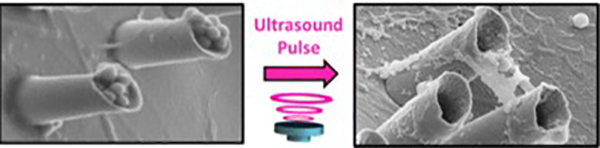
The UCSD scientists successfully fired their nanobullets into artificial tissue, and found that, as predicted in their simulations, those nanobullets were able to penetrate the tissue more deeply.
Going forward, Wang and Esener will be developing this ballistic tool further, so that it is capable of delivering many different kinds of drugs, and even simultaneous delivery of many drugs. It’s also possible such microcannons might prove useful for administering vaccines.
References:
Husseini, G.A. and Pitt, W.G. (2008) “Micells and nanoparticles for ultrasonic drug and gene delivery,” Adv. Drug Deliv. Rev. 60(1): 1137-1152
Soto, F. et al. (2015) “Acoustic microcannons: Towards advanced micro ballistics,” ACS Nano. Published online December 21, 2015.
Images: Soto et al./University of California, San Diego.
